Apostle technologies have been applied in many world-class R&D studies, clinical laboratory settings, and public health response and surveillance. For a complete list of applications citing Apostle technologies, including publications and customer testimonials, see References.
Click the banner below for details of the citing articles.
Apostle technologies have been cited or discussed in the following publications.
2024-2026
2021-2023
Apostle technologies have been cited or used by scientists from the following organizations. Click to read more when there is a publication.
Publications (2026)
106. Earlier postoperative ctDNA detection predicts recurrence and adjuvant therapy benefit in stage II-III colorectal cancer. Zhang, W., Xu, C., Wang, J. et al. npj Precision Oncology. 2026. https://doi.org/10.1038/s41698-026-01497-9
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Detection of postoperative minimal residual disease (MRD) using circulating tumor DNA (ctDNA) accurately predicts cancer recurrence and may even guide adjuvant chemotherapy. Cost-effective and easily implementable assays capable of detecting MRD early (within days after surgery) will drive wider clinical adaptation. We developed a rapid, single-tube multiplex quantitative methylation-specific PCR (mqMSP) assay targeting 10 rigorously screened methylation markers. In a preoperative cohort, mqMSP achieved 100% specificity in 96 controls, with an overall sensitivity of 73.1% for CRC. In a postoperative cohort of 246 stage II-III CRC patients with a long follow-up time (7–96 months, median: 48 months), we demonstrated that ctDNA detection by mqMSP at an early timepoint (median: 5 days after surgery) predicted worse disease-free survival (HR 7.43, P < 0.0001) and overall survival (HR 8.81, P < 0.0001). Benefit from completion of recommended adjuvant therapy was only seen for ctDNA-positive patients, but not for ctDNA-negative patients.
Methods section
Sample Processing
Cell-free DNA (cfDNA) from plasma was isolated with the Apostle MiniMax™ High Efficiency cfDNA Isolation Kit(Apostle; San Jose, CA, USA).
105. Comprehensive molecular characterization of cfDNA as predictive and monitoring biomarkers in advanced gastric cancer receiving immunotherapy. Fang J, Yu Y, Sun Y, Ji M, Jiang Y, Chen X, et al. Journal for ImmunoTherapy of Cancer. 2026;14:e013792. https://doi.org/10.1136/jitc-2025-013792
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background Advanced gastric cancer (aGC) exhibits substantial heterogeneity in response to combination immunotherapy. Circulating cell-free DNA (cfDNA) enables non-invasive profiling of tumor dynamics and may provide biomarkers for response prediction.
Methods We enrolled 94 patients with aGC undergoing combination immunotherapy and assigned them to a discovery set (n=49) and an internal validation set (n=45). Plasma cfDNA was collected pre-treatment and post-treatment and profiled by low-pass whole-genome sequencing and whole-genome bisulfite sequencing in the discovery set, with targeted bisulfite sequencing used in the validation set.
Results The discovery set included 34 responders and 15 non-responders, and the validation set included 30 responders and 15 non-responders. We found that responders showed longer cfDNA, lower cfDNA tumor fraction (median: 0.06 vs 0.01, p<0.001), reduced chromosomal instability (median genomic instability index: 0.026 vs 0.007, p<0.001), and higher global methylation (median: 0.715 vs 0.724, p=0.007). Additionally, the differentially methylated region (DMR) at chr20:25849353-25849490 consistently showed higher methylation in responders in both the discovery (adjusted p=0.006) and validation sets (adjusted p=0.049). A predictor based on this DMR outperformed programmed death-ligand 1 (PD-L1) combined positive score (CPS) with area under the curve (AUCs) of 0.79 (discovery: 95% CI 0.65 to 0.93) and 0.72 (validation: 95% CI 0.54 to 0.91). When integrating PD-L1 CPS with this DMR, the AUCs were 0.81 (discovery: 95% CI 0.67 to 0.95) and 0.75 (validation: 95% CI 0.56 to 0.94), respectively. For on-treatment monitoring, three DMRs increased specifically in responders; among them, increased methylation of chr8:110479193-110480324 and chr8:50891437-50892120 was associated with improved progression-free survival (median: 4.90 vs 11.57 months, p<0.001; median: 5.20 vs 10.20 months, p=0.007).
Conclusion Integrated cfDNA profiling captures immunotherapy-associated molecular dynamics in aGC. A single pretreatment cfDNA methylation marker (chr20:25849353-25849490) improves response prediction beyond PD-L1 CPS and represents a potential predictive biomarker for combination immunotherapy.
Methods section
cfDNA isolation
cfDNA was isolated from 500 µL of plasma using the MiniMax High Efficiency cfDNA Isolation Kit (Apostle, San Jose, California, USA), following the manufacturer’s protocol with a modification for protease digestion.
104. Comprehensive Evaluation of cfRNA Recovery Efficiency Across Tube Types and Competitors Using the Apostle MiniMax® High Efficiency cfRNA Isolation Kit. Beckman Coulter Life Sciences. Application Note. Available online April 2026.
(Application Note by an independent party.)

Introduction
The Apostle MiniMax® High Efficiency cfRNA Isolation Kit is designed to deliver high-quality isolation of cell-free RNAs (cfRNA) ranging from 17 to 1,000 nt, without using phenol or chloroform. This kit is powered by Apostle MiniMax® technology which uses magnetic nanoparticles. This solution ensures high recovery efficiency and supports compatibility with a wide range of downstream applications, including next-generation sequencing (NGS), PCR and other molecular assays. The kit is optimized for samples collected in major blood collection tubes (BCTs), particularly those that preserve RNA integrity during storage, making it ideal for liquid biopsy workflows. By providing reliable cfRNA isolation, the Apostle MiniMax® Kit empowers clinical researchers across diverse therapeutic areas such as oncology, cardiovascular, neuroscience research, prenatal and gestation-related studies, virology and public health.
103. Screening of Hepatocellular Carcinoma in Hepatic Cirrhosis Patients by a Novel Blood-Based Multi-Omics Test. Chen L, Liu J, Long M, Hu P. et al. Technology in Cancer Research & Treatment. (2026). https://doi.org/10.1177/15330338261435022
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Introduction
Hepatocellular carcinoma (HCC) screening in patients with hepatic cirrhosis (HC) relies on ultrasound and alpha-fetoprotein (US + AFP), which has limitations in sensitivity, particularly for early-stage HCC detection. This study aims to evaluate the performance of a novel multi-omics blood test, HCCscreen, with its individual components (methylation, AFP, Des-γ-Carboxy Prothrombin (DCP), mutations) and the standard US + AFP for HCC screening in a hepatic cirrhotic population.
Methods
A total of 5078 patients with known high-risk for HCC were recruited. A prospective screening study was conducted on 650 patients with hepatic cirrhosis identified by ultrasound. Blood samples were collected from all patients before the confirmation of diagnosis by imaging and/or pathological examinations. The performance of HCCscreen, individual markers and US + AFP were calculated and compared. Statistics was performed with Graphpad Prism 5.0.
Results
HCCscreen exhibited a sensitivity of 86.3% at a specificity of 81.3%, with a positive predictive value (PPV) of 28.2% and a negative predictive value (NPV) of 98.6%. The positive likelihood ratio (LR+) was 4.61 and the negative LR (LR-) was 0.17. The positive detection rate (PDR) for all markers increased with more advanced HCC stages, whether Barcelona Clinic Liver Cancer (BCLC) or clinical staging. Among the single-omics, methylation showed the highest PDR, followed by AFP, DCP and mutations. HCCscreen demonstrated superior overall performance with an AUC of 0.87, outperforming individual markers like methylation (AUC = 0.76), AFP (AUC = 0.83), and DCP (AUC = 0.77). Crucially, HCCscreen's PDR was significantly higher than US + AFP in early-stage HCC (BCLC-0 and clinical stage I). Furthermore, while AFP's PDR varied significantly by sex, HCCscreen's performance remained consistent across all demographics. Correlation analysis revealed a significant association only between the HCCscreen score and the methylation score.
Conclusions
The multi-omics approach of HCCscreen significantly enhances early HCC detection in patients with hepatic cirrhosis compared to both its individual components and the current standard of US + AFP. Its robust and consistent performance across patient demographics underscores its potential as a superior tool for population-wide early HCC screening.
Methods and Materials section
DNA Extraction, NGS Library Construction, Targeted Sequencing, AFP and DCP Detection
The methods for DNA extraction, library construction, and targeted sequencing were consistent with those previously reported.10 Firstly, cfDNA was extracted from plasma samples using the Apostle MiniMax cfDNA Isolation Kit (C43468, Apostle)
102. Integrated Liquid Biopsy and Tumor Tissue Genomic Profiling of Appendiceal Cancer: cfDNA Burden, Mutation Landscapes, and Clinical Outcomes. Patel, S., Petrosko, P., Gallo, P.H. et al. Ann Surg Oncol, (2026). https://doi.org/10.1245/s10434-026-19351-4
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
Appendiceal cancer (AC) is a rare malignancy that often presents at advanced stages with significant histological variability influencing clinical outcomes. Precise genomic profiling is essential for accurate diagnosis and personalized patient management. This study interrogated DNA from appendiceal tumor tissue, buffy coat cells, and the cell-free DNA component of plasma using a 523-gene panel for comprehensive genomic profiling (CGP) to identify cancer-related genetic mutations in tumor and blood, evaluate tumor mutation burden, and determine genetic markers associated with histologic grade.
Patients and Methods
A total of 73 patients provided blood samples comprising cell-free DNA (cfDNA) and germline buffy coat cells (bcDNA) for analysis compared with tumor tissues available from 56 of these patients. Concordance of mutations between matched tumor tissue and plasma samples (n = 51) was assessed and tumor-specific and germline variants were classified using OncoKB™ clinical criteria to delineate oncogenic and therapeutically actionable variants [level 1 mutations with U.S. Food and Drug Administration (FDA)-approved therapy]. Additionally, cfDNA concentrations were tested for association with clinical and pathologic features and oncologic outcome including disease-specific (DSS) and progression-free (PFS) survival.
Results
Circulating tumor DNA (ctDNA) from plasma cell-free DNA demonstrated high concordance with tumor genomic profiling, reaching 98.4% concordance [median, interquartile range (IQR) 13.5, 21.5] overall and 85.7% (IQR 64.6, 100) for therapeutically actionable level 1 mutations. Prevalent appendiceal tumor-specific mutations included KRAS proto-oncogene, GTPase (KRAS) (41%), GNAS complex locus (GNAS) (30%), tumor protein p53 (TP53) (30%), and SMAD family member 4 (SMAD4) (29%). Tumor-specific TP53, SMAD4, and spectrin alpha, erythrocytic 1 (SPTA1) mutations strongly correlated with intermediate and high-grade histology, whereas GNAS mutations predominated in low-grade tumors. Germline analysis identified coding mutations shared among this patient cohort in notch receptor 4 (NOTCH4) (55%) and BRCA1 associated RING domain 1 (BARD1) (48%) genes, with zinc finger homeobox 3 (ZFHX3) (29%) and adhesion G protein-coupled receptor A2 (ADGRA2), DNA polymerase epsilon (POLE), and transcription factor 3 (TCF3) mutations (all = 23%) specifically enriched in intermediate and high-grade AC. Both histological grade and cfDNA stratified by concentration tertiles independently predicted progression-free and disease-specific survival. Plasma samples exhibited consistently lower variant allele frequencies than solid tumors, limiting sensitivity for discovery of novel mutations exclusively from plasma.
Conclusions
This study supports integrating comprehensive ctDNA assays into standard diagnostic and treatment pathways for AC using large gene panels. TP53, SMAD4, SPTA1, and GNAS mutations serve as prospective tumor-specific molecular classifiers for histological grade, while germline variants in NOTCH4 and BARD1 may further influence disease biology, with ZFHX3, ADGRA2, POLE, and TCF3 affecting grade stratification. Overall cfDNA concentration may serve as a potential prognostic biomarker in AC.
Patients and Methods section
Purification of Cell‑Free DNA from Plasma for Follow‑Up Studies
Cell-free DNA (cfDNA) purification was performed using the Apostle MiniMax High Efficiency cfDNA Isolation Kit (cat. #A17622-250, Beckman Coulter, Indianapolis, IN).
101. Simultaneous Prenatal cfDNA Screening of Aneuploidy, Recessive Single-Gene Conditions, and Fetomaternal Blood Compatibility. Juexiao Sherry Wang , C J Battey , Kyle Trettin, et al. Clinical Chemistry, hvag005, https://doi.org/10.1093/clinchem/hvag005 Published: 19 February 2026.
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
The provision of prenatal genetic screening in 3 core clinical contexts—recessive conditions, aneuploidy, and serological incompatibility—involves multiple tests across reproductive partners. This complexity limits utilization and impairs adherence to guideline-recommended care, particularly in carrier screening, where male partners are frequently not tested when a female carrier is identified. Here, we describe the analytical validation of a fetomaternal integrated recessive, serological, and trisomy genetic screen (FIRSTGENE), a single assay that harnesses in silico fragment-length trajectory analysis to evaluate all 3 contexts simultaneously, identifying clinically relevant variants in both the mother and the fetus without requiring a paternal sample.
Methods
FIRSTGENE screens singleton pregnancies for mutations in 20 recessive genes; RhD compatibility; aneuploidies in chromosomes 13, 18, 21, X, and Y; and 22q11.2 microdeletion. Each part of the test was individually validated using a relevant subset of plasma samples from a curated collection (478 total samples from 456 patients) and 93 cell-line mixtures digested to resemble maternal and fetal cell-free DNA.
Results
FIRSTGENE demonstrated ≥98.2% sensitivity and ≥99.0% specificity for fetal alleles in recessive-disease genes in plasma and cell lines; 100% sensitivity and specificity for RhD compatibility in plasma; 100% sensitivity and ≥99.8% specificity for fetal chromosomal abnormalities in plasma; and ≥99.9% sensitivity and specificity for maternal alleles in recessive-disease genes in plasma.
Conclusions
FIRSTGENE demonstrated high analytical sensitivity and analytical specificity for each component of the assay. Its capability to generate multiple prenatal screening results from a single blood draw may improve the efficiency and accessibility of prenatal genetic screening.
Materials and Methods section
Molecular Biology Workflow
In the FIRSTGENE targeted sequencing-based molecular workflow (Fig. 1C), plasma was isolated from whole blood collected in Streck tubes; cfDNA was extracted [MiniMax High Efficiency cfDNA Isolation Kit (Apostle)] and prepared into libraries using the xGen cfDNA and FFPE Library Prep Kit (IDT) with unique molecular identifiers (27) (Supplemental Fig. 2A).
100. Mononuclear phagocyte system blockade for enhanced liposome-assisted metabolic glycan labeling and circulating biomarker detection in mice. Pingping Feng, Tingbi Zhao, Rundong He. et al. Nano Research (2026) https://doi.org/10.26599/NR.2026.94908501
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
The rapid clearance of nanoparticles by the mononuclear phagocyte system (MPS) severely compromises nanocarrier delivery efficiency and reduces the isolation yield of circulating biomarkers such as nucleosome-bound cell-free DNA (cfDNA) and exosomes. While liposome-based preconditioning is a promising strategy for transient MPS blockade, effective formulations remain scarce. Here, we report a potent MPS-blocking liposome (the Blocker) identified through an integrated screening strategy combining in vitro Kupffer cell uptake assays with in vivo biodistribution profiling. Notably, even upon co-administration, the Blocker effectively extended the circulation half-life of a widely used FDA‑approved PEGylated liposomal carrier by 2.1 folds, based on which liposome-assisted metabolic glycan labeling was significantly enhanced in a mouse tumor model. Furthermore, liposomal MPS blockade markedly inhibited the clearance of endogenous circulating cfDNA and exosomes, leading to a 6.8-fold increase in recovered cfDNA yield. This work highlighted liposomal blockade as a versatile platform for improving nanoparticle delivery and liquid biopsy sensitivity.
Materials section
Apostle MiniMax high efficiency cfDNA isolation kit (catalogue no. A17830) was purchased from Apostle Bio.
99. Whole blood metagenomic next-generation sequencing in the diagnosis of bloodstream infection in patients with hematological diseases. Xinhao Chai, Xing Zhang, Dongsheng Chen, Dongwen Rong. International Journal of Infectious Diseases. Available online 12 January 2026. https://doi.org/10.1016/j.ijid.2026.108375
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objective
To evaluate the value of cell-free DNA (cfDNA) in plasma and genomic DNA (gDNA) in nucleated cell layer of whole blood samples detected by metagenomic next-generation sequencing (mNGS) in the diagnosis of bloodstream infection in patients with hematological diseases.
Methods
Whole blood samples collected from hematologic patients with suspected bloodstream infections were divided into the plasma and nucleated cell layers. The DNA of plasma and nucleated cell layers was extracted for mNGS. The pathogenic results were compared between whole blood (plasma plus nucleated cell layers) and plasma layer. In addition, the factors influencing the prognosis at discharge were analyzed.
Results
Totally 92 patients were included. The positive rate of mNGS in whole blood was higher than those of the single plasma layer (58.70% vs. 53.26%) and the culture layer (58.70% vs. 17.39%). The consistency of plasma and nucleated cell layers was 57.6%. The proportion of fungi detected in nucleated cell layer was higher than that in plasma layer (30.2% vs. 17.0%). Ten patients had extra pathogens detected in whole blood compared with the single plasma layer, and the positive rate of mNGS increased by 10.87%. gDNA microbe reads and non-host ratios in the extra-detection group were significantly higher than those in the non-extra detection group. cfDNA microbe reads, non-host ratios and microbe percent showed no significant differences between the two groups. The maximum Sequential Organ Failure Assessment (SOFA) score and age in the death group were significantly higher, while cfDNA/gDNA species richness was significantly lower compared with the survival group. The maximum SOFA score and cfDNA Shannon diversity index were found as risk factors for improved prognosis. The maximum SOFA score and cfDNA concentration were combined for the diagnosis of poor prognosis at discharge, with the highest area under the curve of 0.95.
Conclusion
Simultaneous metagenomic sequencing of plasma layer and nucleated cell layer contributes to the detection of pathogens in patients with bloodstream infection. cfDNA detection has a certain significance in predicting the prognosis of patients with bloodstream infection.
Methods section
DNA extraction
After centrifugation, the supernatant was collected into a new 10 mL centrifuge tube and cfDNA was extracted directly with the Apostle MiniMax kit (Apostle,A17622-ACRG-LV) without wall breaking. The precipitate from the bottom of the tube was mixed with 200 uL of the nucleated cell layer and added to the sequenced MP Lysing Matri E tube.
Publications (2025)
98. Circulating plasma EBV DNA: a potential tool to facilitate diagnosis of pediatric Burkitt lymphoma in sub-Saharan Africa. Karena Volesky-Avellaneda, Rena R. Xian, Todd B. Young, Ryan N. Baugher, Martin D. Ogwang, Patrick Kerchan, Steven J. Reynolds, Constance N. Tenge, Pamela A. Were, Robert T. Kuremu, Walter N. Wekesa, Nestory Masalu, Esther Kawira, Stephanie D. Mellott, Heidi E. Lawhorn, Kristen M. Pike, Jennifer Stone, Laura Walsh, Tobias Kinyera, Isaac Otim, Hadijah Nabalende, Herry Dhudha, Leona W. Ayers, Kishor Bhatia, James J. Goedert, Daniel R. Soppet, Stephen M. Hewitt, Hyokyoung G. Hong, Richard F. Ambinder, and Sam M. Mbulaiteye. Blood Global Hematology (2025) Volume 1, Issue 3, December 2025, 100025
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Due to diagnostic delays in sub-Saharan Africa, many pediatric patients with Burkitt lymphoma (BL) perish. A subtype of BL, accounting for 95% of pediatric BL cases in sub-Saharan Africa, is characterized by the presence of Epstein-Barr Virus (EBV). We assessed plasma EBV DNA in 400 BL cases and 400 controls frequency-matched on sex, age, and country enrolled in the Epidemiology of Burkitt lymphoma in East African Children and Minors (EMBLEM) study in Uganda, Kenya, and Tanzania (2010-2016). EBV was measured using digital droplet PCR assay targeting EBV BamH1-W internal repeats, duplexed with RPP30 human housekeeping gene. The study population was predominately male (63% of cases and 64% of controls), with an average age of approximately 7.5 years. EBV DNA was detected in 309 (77.3%) BL cases and 62 (15.5%) controls. The mean plasma EBV DNA levels were 5.00 (standard deviation [SD] 1.63) log10 copies/mL for BL cases versus 1.94 (SD 1.35) for controls (p<0.0001). Excluding 50 BL cases and 61 controls with indeterminate (RPP30 and EBV negative) samples, the maximum sensitivity of plasma EBV DNA detection to discriminate BL cases from controls was 88.3% (95% CI 84.4-91.5%), with 81.7% (77.2-85.7%) specificity, and area under the curve 91.9% (89.7-93.9%). A specificity of 100% was achieved at a threshold of 4.19 log10 EBV copies/mL, which reduced sensitivity to 66.6%. Assay accuracy varied from 83%-87.4% at different thresholds. Testing for EBV DNA could facilitate the recognition of BL earlier in sub-Saharan Africa, a critical step in improving BL cure rates in the region.
Materials and Methods section
Plasma DNA extraction
DNA was extracted from 250 µL aliquots of plasma from previously unthawed plasma sample vials. Plasma aliquots were thawed via heat block at 37°C for five minutes, then transferred to a KingFisher™ Deep-Well 96 Plate (Life Technologies Corporation Carlsbad, CA, USA) on the automated Hamilton Microlab® Prep™ Liquid Handling System (Hamilton Company Reno, NV, USA) where DNA extraction was performed using the Apostle MiniMax™ High Efficiency cfDNA Isolation Kit (Beckman Coulter Inc., Sykesville, MD, USA) with a modified 55°C incubation and reagents and volume scaled down for 250 µL plasma input. Elution incubation was modified from the manufacturer’s protocol to be performed at 55°C.
97. Plasma cell-free DNA methylation markers for detection and prognosis of gastric cancer: A case-control study. Yanan Wang , Longyang Jin, Weiyang He, et al. Chin J Cancer Res. 2025 Oct 30;37(5):851–864. doi: 10.21147/j.issn.1000-9604.2025.05.14
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objective
Plasma cell-free DNA (cfDNA) methylation has shown potential in the detection and prognostic testing of multiple cancers. Here, we comprehensively investigate the performance of cfDNA methylation for gastric cancer (GC) detection and prognosis.
Methods
GC-specific differentially methylated regions (DMRs) were identified by sequencing 56 GC tissues and 59 normal adjacent tissues (NATs). We then performed targeted bisulfite sequencing of cfDNA from 294 GC and 446 non-gastric cancer (NGC) plasma samples, identifying 179 DMRs that overlapped with those in tissue samples. The efficacy of plasma cfDNA methylation markers for GC detection and prognosis was evaluated.
Results
Based on the 179 DMRs overlapping with those in tissue samples, the random forest (RF) model using 28 DMRs achieved an area under the curve (AUC) of 0.998 in the training cohort, whereas further refinement to the top 6 DMRs resulted in an AUC of 0.985. Consistent results were obtained in the validation cohort (28 DMR AUC: 0.985; 6 DMR AUC: 0.988). Support vector machine (SVM) and logistic regression (LR) models also demonstrated robust performance. Additionally, an 11-DMR signature was developed for prognostic prediction, successfully identifying high-risk GC patients with significantly shorter overall survival.
Conclusions
Our study highlights the potential utility of cfDNA methylation markers for both the detection and prognostication of GC.
Materials and Methods section
Plasma DNA extraction
DNA extraction and quality control
Plasma cfDNA extraction was performed utilizing the Apostle MiniMax High-Efficiency cfDNA Isolation Kit (Apostle, A17622CN, Santa Clara, USA).
96. Analysis of urine cell-free DNA copy number and fragment size from healthy individuals. Jufen Wang, et al. Clinica Chimica Acta (2025) In Press. Available online 23 September 2025, 120627 (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objective
This study characterizes urine cell-free DNA (cfDNA) copy number and fragment size in healthy individuals and explores their associations with routine clinical parameters.
Methods
Sixty healthy subjects were enrolled, providing paired blood and urine samples. Six primer pairs targeting nuclear (GAPDH-61/168/241) and mitochondrial DNA (ND1–57/167/240) were designed for absolute qPCR. Optimal urine collection, pre-treatment, and cfDNA detection protocols were evaluated. Correlations between cfDNA characteristics (fragment size and copy number) and clinical parameters (complete blood count, urinalysis, hepatic/renal function biomarkers) were analyzed.
Results
Non-extracted urine retained a higher proportion of fragments <100 bp and > 2000 bp than extracted samples. The optimal pre-treatment involved immediate EDTA addition, centrifugation at 4 °C, and storage at −80 °C. Urine cfDNA comprised short, medium, and long fragments. Cell-free mitochondrial DNA (cf-mtDNA) showed the highest copy numbers in short fragments, decreasing with length, whereas cell-free nuclear DNA (cf-nDNA) peaked in medium fragments. ND1–57 Cq values correlated negatively with neutrophil percentage (P < 0.01) and positively with lymphocyte percentage (P < 0.05). Lymphocyte percentage was moderately correlated with ND1 short fragment (ND1-S, P < 0.01) and weakly with the ND1-S to ND1 medium fragment (ND1-M) ratio (P < 0.05). Absolute lymphocyte count correlated weakly with ND1-S (P < 0.01) and ND1-M (P < 0.05). Neutrophil percentage correlated weakly with ND1-S (P < 0.01) and ND1-S to ND1 long fragment (ND1-L) ratio (P < 0.05).
Conclusion
Urine cfDNA displays three distinct fragment sizes, with cf-mtDNA predominantly found in short fragments and showing stronger associations with physiological parameters than cf-nDNA.
Patients and Methods section
2.3 cfDNA extraction and determination cfDNA was extracted from both plasma and urine samples using the MiniMax™ High-Efficiency cfDNA Isolation Kit (Apostle, USA), following the manufacturer's rapid extraction protocol. Fragment size distribution was analyzed using the Agilent 4150 TapeStation system, in accordance with the manufacturer's instructions.
95. Cell Free DNA, Tumor Molecular Concordance and Clinical Correlates of Cancer Patients Treated in a Large Community Health Care Network. William A. LaFramboise, Patti Petrosko, Phillip H. Gallo, et al. The Journal of Molecular Diagnostics. Volume 27, Issue 9, P882-898, September 2025. DOI: 10.1016/j.jmoldx.2025.05.007 (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Blood collection, plasma processing and cell free DNA (cfDNA) purification was optimized to capture circulating tumor DNA (ctDNA) without blood cell background DNA among 874 cancer patients. cfDNA comprised predominantly mononucleosomal fragments (n=874; x¯ ± S.D. = 166 ± 5 bp) that generated comparably sized sequencing reads (x¯ ± S.D. = 162 ± 25 bp). Despite a vast range of cfDNA concentrations (0.50 to 1132.9 ng/ml) across 21 tumor types, matched tumor and blood specimens (n = 430 patients) revealed high concordance for coding (Med = 97%) and clinical oncogenic mutations (Med = 88% concordance). Therapeutically actionable mutations were identified in 233 patients by both assays while 126 patients had oncogenic mutations without an established pharmacotherapeutic agent. An additional 48 patients (11%) had actionable mutations detected only in cfDNA assays while 23 patients (5%) had mutations in tumor only. Concordance was high in both prevalent (lung, breast, colon) and rare tumors (appendiceal, sarcoma). Cell free DNA levels from diagnostic blood specimens were a strong indicator of patient survival duration independent of age, sex, tumor type and stage, demonstrative of a potentially important role as a prognostic biomarker. Mutations in established oncogenes and tumor suppressors were readily detectable across all tumor types in ctDNA indicating a diagnostic role for cfDNA from blood extending beyond the identification of companion therapeutics to patient screening and monitoring.
Materials and Methods section
Purification of Cell Free DNA from Plasma. Frozen plasma was thawed at room temperature (60 min) and the volume adjusted as needed (1X PBS, pH 7.4; cat. #10010-023, ThermoFisher) for purification of cell-free DNA (cfDNA) using the Apostle MiniMax High Efficiency cfDNA Isolation Kit (cat. #A17622-250, Beckman, Indianapolis, IN).
94. Integrated analysis of oral rinse-derived and plasma circulating tumour DNA for mutation profiling and outcome prediction with oral squamous cell carcinoma. Zhuo Chen, Lihuang Chen, Shuaize Li, et al. npj Precision Oncology. volume 9, Article number: 183 (2025) (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
This study evaluates the potential of oral rinse-derived and plasma circulating tumour DNA (ctDNA) in HPV-negative oral squamous cell carcinoma (OSCC), where early recurrence occurs in a significant proportion of patients, contributing to poor prognosis. Analysis of paired tissue, oral rinse, and plasma samples from 123 patients revealed ctDNA detection rates of 94.3% in oral rinse and 80.5% in plasma samples. Combined testing improved mutation detection sensitivity to 48.6%. A machine learning model integrating seven mutated genes (TP53, TERT, IKZF1, EP300, MYC, EGFR, PIK3CA) and clinical factors demonstrated robust prediction of recurrence (validation AUC: 0.854) and survival outcomes. Integration of pretreatment plasma ctDNA status further enhanced predictive performance. In longitudinal analysis, ctDNA detected recurrence approximately four months before clinical manifestation. These findings suggest that integrated ctDNA analysis offers improved mutation profiling and outcome prediction, potentially enabling earlier interventions in OSCC.
Methods section
Plasma samples were collected using Apostle tubes. All samples were transported at 2–8 °C and subsequently stored at -80 °C until analysis.
93. Novel Viral Sequences in a Patient with Cryptogenic Liver Cirrhosis Revealed by Serum Virome Sequencing. Xiaoan Zhang, Ida X. Fan, Yanjuan Xu, Jody Rule, Long Ping Victor Tse, Mahmoud Reza Pourkarim, William M. Lee, Adrian M. Di Bisceglie and Xiaofeng Fan. Viruses 2025, 17(6), 812. (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Clinical studies indicate the etiology of liver disease to be unknown in 5% to 30% of patients. A long-standing hypothesis is the existence of unknown viruses beyond hepatitis A through E virus. We conducted serum virome sequencing in nine patients with cryptogenic liver disease and identified eight contigs that could not be annotated. One was determined to be a contaminant, while two of seven contigs from an individual (Patient 3) were validated by reverse transcription and polymerase chain reaction (RT-PCR) and Sanger sequencing. The possibility of contamination was completely excluded through PCR, with templates extracted using different methods from samples taken at different time points. One of the contigs, Seq260, was characterized as negative-sense single-stranded DNA via enzymatic digestion and genome walking. Digital-droplet PCR revealed the copy number of Seq260 to be low: 343 copies/mL. Seq260-based nested PCR screening was negative in 200 blood donors and 225 patients with liver disease with/without known etiologies. None of the seven contigs from Patient 3 was mapped onto 118,713 viral metagenomic data. Conclusively, we discovered seven unknown contigs from a patient with cryptogenic liver cirrhosis. These sequences are likely from a novel human virus with a negative-sense, linear single-stranded DNA genome.
Materials and Methods section
2.8. Detection of Seq260 in the Screened Cohort
Total DNA was extracted from serum samples in the screened cohort using Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit (Apostle, Pleasanton, CA, USA). An aliquot of 2.5 ng of extracted DNA was used as the input for the nested PCR with primers designed based on Seq260 (Table 2). The product of the PCR was visualized on agarose gel. Suspected amplicons were gel-purified and subjected to Sanger sequencing.
92. Combined genomic profiling of cell-free DNA (cfDNA) and circulating tumor cell DNA (ctcDNA) in S1802, a prospective phase 3 trial for metastatic prostate cancer (mHSPC). Daniel Bsteh, Jonathan Buckley, Scott Mitchell, et al. Journal of Clinical Oncology. Volume 43, Number 16_suppl. Meeting Abstract: 2025 ASCO Annual Meeting. May 28, 2025 (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background: Parallel analysis of cfDNA and ctcDNA can yield expanded and complementary molecular insights. We developed HERCULES, a prostate cancer specific targeted amplicon sequencing panel capable of assessing single nucleotide variants (SNVs) and copy number variants (CNVs) in cfDNA and single CTCs obtained concurrently from a single tube of blood. Here we present results from the first 35 patients analyzed at multiple time points in S1802, an NCTN/SWOG randomized prospective phase 3 trial of standard systemic therapy +/- definitive treatment of the primary tumor in newly diagnosed mHSPC.
Methods: Blood was collected in Streck DNA preservative tubes at 4 prespecified time points: registration, randomization, post-definitive therapy, and progression. Plasma and single CTCs were isolated using the RareCyte platform, and DNA was extracted using the Apostle MiniMax cfDNA extraction kit (Beckman) or Single Cell Lysis Kit (Thermo Fisher). The HERCULES AmpliSeq-HD panel includes 35 prostate cancer -relevant genes. SNVs and CNVs were called using Ion Reporter Software (Thermo Fisher) and filtered for variants associated with clonal hematopoiesis of indeterminate significance (CHIP).
Results: A total of 85 samples were obtained from 35 patients at up to 4 different time points. CTCs were detected in 22(61%) patients, with median 3 CTCs/7.5ml (range 1-986). SNV limit of detection varied with input DNA, from 0.16% at >20 ng input to 0.91% with 1-5 ng input. Fewer than 5% of all SNVs were censored due to potential CHIP. Across all times points, SNVs and CNVs were detected in 32 and 20 patients, respectively. SNVs were detected both in cfDNA and in ctcDNA. Known prostate cancer driver variants were observed in AR, CTNNB1, FOXA1, SF3B1, TP53 and others. Recurrent CNVs were observed in AR, AR enhancer, FOXA1, CDK4, MYC and CHD1. In general, the 4th time point, representing transition to mCRPC, had twice the number of alterations as earlier mHSPC time points.
Conclusions: Analysis of the first 35 patients sequenced at multiple time points in S1802 demonstrates the feasibility of concurrent, same-sample genomic profiling of cfDNA and ctcDNA in a multi-center prospective phase 3 NCTN setting. The HERCULES sequencing workflow enables parallel processing of cfDNA and single-cell ctcDNA, with LOD that exceeds that of hybrid capture panels at these input levels. Presence of concordant and distinct alterations in cfDNA and CTCs is consistent with findings from prior small studies and underscores the potential value of analyzing both of these liquid biopsy components.
91. The GENESTAR manual for biospecimen collection biobanking and omics data generation from commercial space missions. Aparna Krishnavajhala, Marie-Claude Gingras, Emmanuel Urquieta, et al. npj Microgravity volume 11, Article number: 16 (2025). (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
The surge in commercial and civilian spaceflight enables the systematic and longitudinal, large-scale biospecimen collection to understand the prospective effects of space travel on human health. The Genomics and Space Medicine (Space Omics) project at BCM-HGSC involves a comprehensive biospecimen collection plan from commercial/private space flight participants. The manuscript addresses the critical gaps in the biospecimen collection process including details of the informed consent process, a provision for subjects to obtain custom CLIA-WGS reports, a data dictionary and a LIMS enabled biobank. The manuscript also discusses the biospecimens collection, processing methodologies and nucleic acid suitability for Omics data generation. Results from Axiom-2 mission where, 339 biospecimens were collected using ‘Genomic Evaluation of Space Travel and Research (GENESTAR)’ manual, at two different sites, showed that 98% of the blood samples and 91.6% of the non-blood samples passed the QC requirements for Omics assays, underscoring the reliability and effectiveness of the GENESTAR manual.
(Methods section)
Nucleic Acid Extractions from blood samples
Cell-free DNA was extracted from the BCT tube using the Apostle MiniMax High-efficiency cfDNA Isolation kit (VWR, cat# 76409-757), as per the manufacturer’s instructions.
90. Integrating Plasma Cell-Free DNA Fragment End Motif and Size with Genomic Features Enables Lung Cancer Detection. Tae-Rim Lee, Jin Mo Ahn, Junnam Lee , et al. Cancer Research (2025) 85 (9): 1696–1707. MAY 02 2025. (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Early detection of lung cancer is important for improving patient survival rates. Liquid biopsy using whole-genome sequencing of cell-free DNA (cfDNA) offers a promising avenue for lung cancer screening, providing a potential alternative or complementary approach to current screening modalities. Here, we aimed to develop and validate an approach by integrating fragment and genomic features of cfDNA to enhance lung cancer detection accuracy across diverse populations. Deep learning–based classifiers were trained using comprehensive cfDNA fragmentomic features from participants in multi-institutional studies, including a Korean discovery dataset (218 patients with lung cancer and 2,559 controls), a Korean validation dataset (111 patients with lung cancer and 1,136 controls), and an independent Caucasian validation cohort (50 patients with lung cancer and 50 controls). In the discovery dataset, classifiers using fragment end motif by size, a feature that captures both fragment end motif and size profiles, outperformed standalone fragment end motif and fragment size classifiers, achieving an area under the curve (AUC) of 0.917. The ensemble classifier integrating fragment end motif by size and genomic coverage achieved an improved performance, with an AUC of 0.937. This performance extended to the Korean validation dataset and demonstrated ethnic generalizability in the Caucasian validation cohort. Overall, the development of a deep learning–based classifier integrating cfDNA fragmentomic and genomic features in this study highlights the potential for accurate lung cancer detection across diverse populations.
Significance: Evaluating fragment-based features and genomic coverage in cell-free DNA offers an accurate lung cancer screening method, promising improvements in early cancer detection and addressing challenges associated with current screening methods.
(Materials and Methods section)
cfDNA extraction, library preparation, and sequencing
cfDNA was isolated from 0.4 mL of plasma using the Apostle MiniMax Kit (Beckman Coulter) following the manufacturer’s guidelines.
89. Trauma-induced inflammation: From targeted therapy for muscle repair to B cells in bone healing. Ort, Melanie-Jasmin. Inaugural-Dissertation, Doctor rerum naturalium (Dr. rer. nat.) 2025-05-13. Freie Universität Berlin. (Download PDF)
(Note: Apostle MiniMax technology is used in this Dissertation.)
 Abstract
Abstract
Inflammation, followed by an anti-inflammatory phase mediated by immune cells, is essential for tissue healing. Prolonged inflammation caused by severe injuries can impair regeneration. Elderly patients suffering from major trauma, such as a hip fracture, are at high risk for delayed healing due to excessive inflammation. This leads to muscle loss and fibrosis post-surgery, which reduces the quality of life and increases mortality in this cohort. Cell therapy could support patients in their regenerative process. This thesis investigates the immunomodulatory effects of placenta derived stroma cell (PLX-PAD) therapy on muscle repair in a Phase III study. PLX-PAD promotes the differentiation and migration of muscle precursor cells in vitro. Their unique properties counteract pro-inflammatory stimuli from activated immune cells. Injection of PLX-PAD during surgery resulted in improved muscle strength and volume, highlighting their therapeutic potential. Fracture healing shares similarities with muscle regeneration, where prolonged inflammation can lead to delayed healing and non-union of bones. This process is influenced by the individual immune cell composition in the local hematoma, bone marrow, and peripheral blood. This work reveals how fracture hematoma is a distinct and transient tissue with diverse immune cell subsets, including B cells. B cells appear to play a role in fracture healing, but their role in trauma has not been characterized well. B cells in the fracture situation shed the complement receptor CR2/CD21, likely triggered by cell-free DNA released from damaged cells. Fracture hematoma also induced the dsDNA Toll-like receptor 9 in vitro. This activation may help clear cellular debris and promote an anti-inflammatory environment. As described in the literature, the expected antibody response upon CD21/TLR9 activation did not yield conclusive results in this study and corresponding cell cultures. Due to the unpredictable nature of trauma, patient-specific research is challenging. A bone and bone marrow organ-on-a-chip system was developed to replicate the immune composition of patients and predict clinical outcomes, such as the impact of metal implant debris. This system will be further refined to study fracture healing over time while preserving individual donor variability. In summary, this thesis emphasizes the importance of modulating the local microenvironment for musculoskeletal regeneration by interacting with pro-inflammatory immune cells and fostering an anti-inflammatory milieu necessary for successful healing.
(Methods section)
2.7 Cell-free DNA Isolation from BM, blood and FH supernatant
Apostle MiniMax™ High Efficiency cfDNA Isolation Kit (Beckmann Coulter, Washington D.C., USA) was used as described in the manufacturer's protocol.
88. Enzymatic profiling of cfDNA methylation for detection and monitoring of lung cancer. Abed Agbarya, Noa Gilat, Yael Michaeli, Jasline Deek, Assaf Grunwald, Suheil Artul, Rasha Khoury, Michael Peled, Yuval Ebenstein. medRxiv, May 08, 2025. https://doi.org/10.1101/2025.05.08.25327181
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
We present a highly sensitive, low-cost approach for detecting lung cancer and monitoring response to therapy, based on sequencing-free detection of methylation biomarkers in cell-free DNA. An engineered methyltransferase is used to fluorescently label CpG sites. When applied to bisulfite-treated, PCR-amplified cell-free DNA, fluorescent reporters attach to all originally methylated sites, which can then be read on a standard hybridization microarray. In a proof-of-concept study involving 60 blinded participants, we distinguished cancer patients from healthy individuals with both sensitivity and specificity exceeding 90 %.
(Materials and Methods section)
Sample preparation, labeling and hybridization
Whole blood samples were collected in Streck tubes (STRECK). Plasma was separated and cfDNA was extracted with Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit according to manufacturer’s instructions. Following DNA extraction.
87. Analysis of the clinical application value of cfDNA methylation and fragmentation in early diagnosis of esophageal cancer. Xin Liu, Chen Liang, Lingwen Ding, et al. Genomics. Volume 117, Issue 3, May 2025, 111034 (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
This study explores the clinical value of cfDNA methylation and fragmentation for the early diagnosis of esophageal cancer using liquid biopsy.
Methods
Whole genome bisulfite sequencing and low-pass whole genome sequencing were utilized to detect cfDNA biomarkers, comparing 30 esophageal cancer patients with 10 healthy controls.
Results
Significant differences in cfDNA methylation and fragmentation were observed between cancerous and non-cancerous samples (p < 0.05). A volcano plot identified 822 differentially methylated markers (817 upregulated, 5 downregulated), with SOX17, SOX1, ZNF382, ZNF667-AS1, and TFPI2 highly associated with esophageal cancer. Fragmentation markers (EDM, FSD, FSR, TFBS, CNV) showed 95 % specificity and sensitivity, with EDM demonstrating the best performance.
Conclusion
Our study highlights the clinical potential of cfDNA methylation and fragmentation biomarkers for the early diagnosis of esophageal cancer.
(Materials and Methods section)
Sample collection, storage, and extraction
Peripheral blood samples (2 tubes, totaling 20 mL) were collected from each participant in Apostle tubes for the detection of methylation and fragmentation markers.
86. Longitudinal genomic profiling of chemotherapy-related CHIP variants in patients with ovarian cancer. Sara Corvigno, Jun Yao, Amma Asare, et al. Front Oncol. 2025 Apr 29;15:1538446. doi: 10.3389/fonc.2025.1538446 (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Introduction
Clonal hematopoiesis (CH) is characterized by the presence of hematopoietic stem cells (HSCs) with the potential of clonally expanding and giving rise to hematological malignancies. Clonal hematopoiesis of indeterminate potential (CHIP) is the outgrowth of a single HSC clone with an acquired somatic mutation in the absence of hematological abnormalities. CHIP variants occur with a variant allele frequency (VAF) of at least 2% in peripheral blood. This definition does not account for less frequent mutations that give rise to hematopoietic clones. Previous studies indicate an association between CH and secondary hematologic malignancies in cancer patients who receive chemotherapy.
Methods
To discover novel candidate CHIP mutations, including those with extremely low VAFs, we performed an in-depth characterization of low-frequency CHIP variants in a highly selected group of patients with high-grade serous ovarian cancer (HGSC) before and after neoadjuvant chemotherapy (NACT). We performed comprehensive ultra-high-depth whole-exome sequencing of circulating free DNA (cfDNA) and matched white blood cell (WBC) DNA from pre- (n=9) and post-NACT (n=9) samples from HGSC patients who had excellent response (ER; n=4) or poor response (PR; n=5) to NACT.
Results
Variants present in both the WBC DNA and cfDNA from a patient were considered candidate CHIP variants. We identified 93,088 candidate CHIP variants in 13,780 genes. Compared with pre-NACT samples, post-NACT samples tended to have fewer CHIP mutations with VAFs of less than 5%, which may reflect the negative selective pressure of chemotherapy on rare hematopoietic clones. Finally, we identified CHIP variants in tumor samples matched to the liquid biopsies.
Discussion
Our innovative sequencing approach enabled the discovery of a large number of novel low-frequency candidate CHIP mutations, whose frequency and composition are affected by chemotherapy, in the cfDNA of patients with HGSC. The CHIP variants that were enriched after chemotherapy, if validated, might become essential predictive markers for therapy-related myeloid neoplasia.
(Materials and Methods section)
Isolation of cfDNA and WBC DNA and sequencing
cfDNA was extracted using the MiniMax High Efficiency Cell-Free DNA Isolation Kit (#A17622-50; Apostle) and then quantified using the Quant-iT PicoGreen dsDNA Assay Kit; ...
85. Diagnostic and prognostic potential of cell-free RNAs in cerebrospinal fluid and plasma for brain tumors. Jinyong Huang, Jinxia Zhou, Jun Wang, et al. npj Precision Oncology. 9, Article number: 123 (2025) (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Systematic assessment of the clinical applicability of cell-free RNAs (cfRNAs), which includes broader RNA categories beyond microRNAs, for patients with brain tumors remains largely unexplored due to the lack of sensitive profiling technologies. Our study endeavors to bridge this gap by utilizing an optimized cell-free transcriptome profiling technique that we have recently developed. We comprehensively profiled the cell-free transcriptome in plasma and cerebrospinal fluid (CSF) samples from a total of 85 patients with glioma, meningioma, or tumor-free central nervous system diseases. We identified 16 cfRNA signatures in CSF with robust performance in brain tumor detection (test set AUC = 0.94; validation set AUC = 1). The integration of CSF and plasma-derived cfRNAs outperformed individual analyses using either CSF or plasma candidates for the classification of glioma (test set AUC = 0.94; validation set AUC = 0.85) and meningioma (test set AUC = 0.92; validation set AUC = 0.83). Additionally, we identified 33 CSF and 3 plasma cfRNAs with prognostic significance for postoperative patient outcomes. Multivariate analysis showed that cfRNA-based risk scores (Hazard ratio=9.9) outperformed traditional risk factors in predicting recurrence-free survival. Importantly, our findings in liquid biopsies are consistent with results from primary tumor tissues. By delving into the diagnostic and prognostic implications of cfRNA signatures in CSF and plasma, our study paves the way for improved diagnostic precision and personalized therapeutic interventions for brain tumor patients.
(Methods section)
cfRNA extraction, library preparation, and sequencing
The extraction of cfRNA from 400 μl CSF samples was conducted using the RNAiso Plus total RNA extraction reagent (TaKaRa), following the manufacturer’s protocol. For the plasma samples, cfRNA extraction was carried out on 200 μl aliquoted plasma utilizing the Apostle MiniMax™ High Efficiency cfRNA Isolation Kit (Apostle) in accordance with the manufacturer’s protocol.
84. Genomic alterations of marine yeast Scheffersomyces spartinae under spontaneous and mutagenic conditions. Yu-Ting Bai, Awkash Sharma, Qian Xiang, et al. BMC Genomics. volume 26, Article number: 297 (2025) (Download PDF)
(Note: Apostle MiniGenomics technology is used in this study.)
 Abstract
Abstract
Background
Understanding the mechanisms of genetic evolution in marine yeasts is essential for their ecological and biotechnological applications. Scheffersomyces spartinae, an ascomycetous yeast species, characterized by its remarkable robustness and carbon source utilization capability, has garnered significant attention for its biotechnological potential.
Results
In this study, we investigated the spontaneous and induced genomic alterations of the marine yeast S. spartinae under various conditions. Through mutation accumulation experiments combined with whole-genome sequencing, we revealed that the rates of spontaneous single nucleotide variations and small insertions and deletions were 6.3 × 10⁻¹¹ and 1.4 × 10⁻¹¹ per base pair per cell division, respectively, in S. spartinae. The predominant type of base substitution was C-to-T or G-to-A, likely induced by cytosine deamination. Template slippage during DNA replication emerged as the primary cause of small InDels. 50 J/m2 UV treatment elevated the SNV rate by 124-fold, with C-to-T substitutions occurring at the 5’-TC-3’ motif and T-to-C substitutions at the 5’-TT-3’ motif being the most prominent features. Exposure to 50 µg/mL Zeocin resulted in 76-fold and 71-fold increases in the rates of SNVs and InDels, respectively, with frequent T-to-A mutations and T deletions occurring at the 5’-GT-3’ motifs. Heat stress at 37 °C increased the SNVs and InDels rates to 1.4 × 10⁻¹⁰ and 7.5 × 10⁻¹¹ per base pair per cell division. Notably, this study demonstrated that large deletions and duplications (> 1 kb) and aneuploidies are less likely to occur in S. spartinae compared to other yeast species, suggesting that this organism is less tolerant to large-scale genomic alterations. In contrast, we observed a marked decrease in rDNA copy numbers when S. spartinae cells were cultivated at elevated temperature conditions. This finding indicates that variations in rDNA copy numbers might act as an adaptive strategy for yeasts in response to fluctuating temperatures.
Conclusions
Our findings provide novel insights into the patterns and genetic mechanisms underlying genomic evolution in yeasts.
(Materials and Methods section)
Genome sequencing of S. spartinae isolates
The genomic DNA (gDNA) of yeast cells were extracted using the Genomic DNA extraction kit (Apostle MiniGenomics),...
83. Digital PCR Assay Utilizing In-Droplet Methylation-Sensitive Digestion for Estimation of Fetal cfDNA From Plasma. Richard Dannebaum, Olga Mikhaylichenko, David Siegel, Chenyu Li, Eric Hall, Severine Margeridon, Monica Herrera, Kristin Loomis, Thea Riel, Madhumita Ramesh, Maria Gencoglu, Nathan Hendel, Anthony Henriquez, Nyari Dzvova, Raymond-John Abayan, Xinhua Lin, Martin Chavez, Nazeeh Hanna. Prenatal Diagnosis, March 16, 2025 doi: https://doi.org/10.1002/pd.6774. (Download PDF)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objective Recent guidelines suggest that non-invasive prenatal screening (NIPS) should be offered to all patients with singleton and twin pregnancies. Accurate determination of fetal fraction in cell-free DNA (cfDNA) is vital for reliable NIPS outcomes. We propose a methylation-based approach using droplet digital PCR (ddPCR) and methylation-sensitive restriction enzyme (MSRE) digestion for fetal fraction quantification as an affordable and fast solution.
Method Following biomarker discovery using early pregnancy placental genomic DNA (gDNA) and cfDNA from non-pregnant female individuals, we designed assays targeting MSRE-compatible regions based on contrasting methylation patterns between maternal and fetal cfDNA. We established a proof-of-concept ddPCR workflow on the Bio-Rad Droplet Digital PCR QX600 instrument.
Results Testing the fetal fraction assay multiplex on 137 prospective clinical samples demonstrated high concordance with NGS results for both female and male pregnancies as well as with chromosome Y-based calculations for samples with a male fetus. Reproducibility analysis indicated lower variability compared to previously reported NGS performance.
Conclusion This study showcases the potential of this novel, 6-color, high-multiplex methylation ddPCR panel for accurate measurement of fetal fraction in cfDNA samples. It presents opportunities to integrate such methodology as a standalone measurement to assess the quality of samples undergoing NIPS.
(Methods section)
2.2.2 | Cell‐Free DNA Sample Processing
Libraries from cfDNA were prepared using cell-free DNA extracted from 2 mL of plasma using the Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit (Apostle Inc.).
82. Intestinal microbiota as biomarkers for different colorectal lesions based on colorectal cancer screening participants in community. Gairui Li, Dan Zhao, Binfa Ouyang, et al. Front. Microbiol. , 06 February 2025. Volume 16 - 2025 | https://doi.org/10.3389/fmicb.2025.1529858. (Download PDF)
(Note: Apostle MinoGenomics technology is used in this study.)
 Abstract
Abstract
Introduction: The dysregulation of intestinal microbiota has been implicated in the pathogenesis of colorectal cancer (CRC). However, the utilization of intestinal microbiota for identify the lesions in different procedures in CRC screening populations remains limited.
Methods: A total of 529 high-risk individuals who underwent CRC screening were included, comprising 13 advanced adenomas (Aade), 5 CRC, 59 non-advanced adenomas (Nade), 129 colon polyps (Pol), 99 cases of colorectal inflammatory disease (Inf), and 224 normal controls (Nor). 16S rRNA gene sequencing was used to profile the intestinal microbiota communities. The Gut Microbiota Health Index (GMHI) and average variation degree (AVD) were employed to assess the health status of the different groups.
Results: Our findings revealed that the Nor group exhibited significantly higher GMHIs and the lowest AVD compared to the four Lesion groups. The model incorporating 13 bacterial genera demonstrated optimal efficacy in distinguishing CRC and Aade from Nor, with an area under the curve (AUC) of 0.81 and a 95% confidence interval (CI) of 0.72 to 0.89. Specifically, the 55 bacterial genera combination model exhibited superior performance in differentiating CRC from Nor (AUC 0.98; 95% CI, 0.96-1), the 25 bacterial genera combination showed superior performance in distinguishing Aade from Nor (AUC 0.95). Additionally, the 27 bacterial genera combination demonstrated superior efficacy in differentiating Nade from Nor (AUC 0.82). The 13 bacterial genera combination exhibited optimal performance in distinguishing Inf from Nor (AUC 0.71).
Discussion: Our study has identified specific microbial biomarkers that can differentiate between colorectal lesions and healthy individuals. The intestinal microbiota markers identified may serve as valuable tools in community-based CRC screening programs.
(Materials and Methods section)
DNA extraction, library construction and sequencing
Genomic DNA was extracted from stool samples utilizing the Apostle MiniGenomics Genomic DNA Extraction according to the manufacturer’s instructions.
81. Highly specific multiplex DNA methylation detection for liquid biopsy of colorectal cancer. Dewen Zhu, Jinlei Li, Wenwen Zhang , Clinica Chimica Acta (2025) 565: 120026, doi: https://doi.org/10.1016/j.cca.2024.120026
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
Circulating tumor DNA (ctDNA) has emerged as a useful biomarker for cancer detection and prognosis. In this study, we developed a strategy for developing a highly specific multiplex qPCR assay to detect methylated ctDNA in the blood of colorectal cancer (CRC) patients and investigated the potential use for the detection and prognosis of CRC.
Methods
Bisulfite conversion and amplicon sequencing were used to confirm potential CRC-specific DNA methylation markers. The selected DNA methylation candidates were validated by qMSP. The six best-performing markers were used to develop a new single-tube multiplex quantitative methylation-specific PCR assay (mqMSP). The mqMSP assay was applied to analyze plasma samples from 114 CRC patients, 47 patients with advanced adenoma, 45 patients with benign polyps, and 57 healthy controls. The clinical performance of the assay and associations with clinical outcomes were assessed.
Results
Six DNA methylation biomarkers were confirmed to be specifically hypermethylated in CRC tumor tissues. The newly developed mqMSP assay detected CRC with extremely high specificity (specificity of 98.2 %, with sensitivity of 67.5 %). The detection rate of ctDNA was significantly correlated with tumor size and clinical stage, with ctDNA methylation levels in the blood markedly increased with larger tumor size, poor differentiation, and advanced stage. Moreover, high preoperative methylated ctDNA level was associated with worse recurrence-free survival and overall survival.
Conclusion
We provided a strategy for identification of multiple highly-specific DNA methylation markers for designing multiplex DNA methylation assays for liquid biopsies of CRC. The newly developed assay has potential for CRC early detection, and prognosis.
(Materials and Methods section)
For plasma samples, 2-5 mL plasma was extracted using the Apostle MiniMax High-Efficiency cfDNA Isolation Kit (Apostle).
Publications (2024)
80. A shared neoantigen vaccine combined with immune checkpoint blockade for advanced metastatic solid tumors: phase 1 trial interim results. Amy R. Rappaport, Chrisann Kyi, Monica Lane, et al. Nature Medicine 30, pages 1013–1022 (2024)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Therapeutic vaccines that elicit cytotoxic T cell responses targeting tumor-specific neoantigens hold promise for providing long-term clinical benefit to patients with cancer. Here we evaluated safety and tolerability of a therapeutic vaccine encoding 20 shared neoantigens derived from selected common oncogenic driver mutations as primary endpoints in an ongoing phase 1/2 study in patients with advanced/metastatic solid tumors. Secondary endpoints included immunogenicity, overall response rate, progression-free survival and overall survival. Eligible patients were selected if their tumors expressed one of the human leukocyte antigen-matched tumor mutations included in the vaccine, with the majority of patients (18/19) harboring a mutation in KRAS. The vaccine regimen, consisting of a chimp adenovirus (ChAd68) and self-amplifying mRNA (samRNA) in combination with the immune checkpoint inhibitors ipilimumab and nivolumab, was shown to be well tolerated, with observed treatment-related adverse events consistent with acute inflammation expected with viral vector-based vaccines and immune checkpoint blockade, the majority grade 1/2. Two patients experienced grade 3/4 serious treatment-related adverse events that were also dose-limiting toxicities. The overall response rate was 0%, and median progression-free survival and overall survival were 1.9 months and 7.9 months, respectively. T cell responses were biased toward human leukocyte antigen-matched TP53 neoantigens encoded in the vaccine relative to KRAS neoantigens expressed by the patients’ tumors, indicating a previously unknown hierarchy of neoantigen immunodominance that may impact the therapeutic efficacy of multiepitope shared neoantigen vaccines. These data led to the development of an optimized vaccine exclusively targeting KRAS-derived neoantigens that is being evaluated in a subset of patients in phase 2 of the clinical study. ClinicalTrials.gov registration: NCT03953235.
Abstract Therapeutic vaccines that elicit cytotoxic T cell responses targeting tumor-specific neoantigens hold promise for providing long-term clinical benefit to patients with cancer. Here we evaluated safety and tolerability of a therapeutic vaccine encoding 20 shared neoantigens derived from selected common oncogenic driver mutations as primary endpoints in an ongoing phase 1/2 study in patients with advanced/metastatic solid tumors. Secondary endpoints included immunogenicity, overall response rate, progression-free survival and overall survival. Eligible patients were selected if their tumors expressed one of the human leukocyte antigen-matched tumor mutations included in the vaccine, with the majority of patients (18/19) harboring a mutation in KRAS. The vaccine regimen, consisting of a chimp adenovirus (ChAd68) and self-amplifying mRNA (samRNA) in combination with the immune checkpoint inhibitors ipilimumab and nivolumab, was shown to be well tolerated, with observed treatment-related adverse events consistent with acute inflammation expected with viral vector-based vaccines and immune checkpoint blockade, the majority grade 1/2. Two patients experienced grade 3/4 serious treatment-related adverse events that were also dose-limiting toxicities. The overall response rate was 0%, and median progression-free survival and overall survival were 1.9 months and 7.9 months, respectively. T cell responses were biased toward human leukocyte antigen-matched TP53 neoantigens encoded in the vaccine relative to KRAS neoantigens expressed by the patients’ tumors, indicating a previously unknown hierarchy of neoantigen immunodominance that may impact the therapeutic efficacy of multiepitope shared neoantigen vaccines. These data led to the development of an optimized vaccine exclusively targeting KRAS-derived neoantigens that is being evaluated in a subset of patients in phase 2 of the clinical study. ClinicalTrials.gov registration: NCT03953235.
(Methods section)
cfDNA was extracted from the entire plasma volume of a single draw using the Apostle MiniMax cfDNA Isolation kit (ApostleBio) and quantified using the Qubit 1× dsDNA High Sensitivity Assay (Thermo Fisher Scientific).
79. Tumor- and circulating-free DNA methylation identifies clinically relevant small cell lung cancer subtypes. Simon Heeke, Carl M. Gay, Marcos R. Estecio, et al. Cancer Cell Volume 42, Issue 2, Pages 225-237.e5 (2024)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Small cell lung cancer (SCLC) is an aggressive malignancy composed of distinct transcriptional subtypes, but implementing subtyping in the clinic has remained challenging, particularly due to limited tissue availability. Given the known epigenetic regulation of critical SCLC transcriptional programs, we hypothesized that subtype-specific patterns of DNA methylation could be detected in tumor or blood from SCLC patients. Using genomic-wide reduced-representation bisulfite sequencing (RRBS) in two cohorts totaling 179 SCLC patients and using machine learning approaches, we report a highly accurate DNA methylation-based classifier (SCLC-DMC) that can distinguish SCLC subtypes. We further adjust the classifier for circulating-free DNA (cfDNA) to subtype SCLC from plasma. Using the cfDNA classifier (cfDMC), we demonstrate that SCLC phenotypes can evolve during disease progression, highlighting the need for longitudinal tracking of SCLC during clinical treatment. These data establish that tumor and cfDNA methylation can be used to identify SCLC subtypes and might guide precision SCLC therapy.
Abstract Small cell lung cancer (SCLC) is an aggressive malignancy composed of distinct transcriptional subtypes, but implementing subtyping in the clinic has remained challenging, particularly due to limited tissue availability. Given the known epigenetic regulation of critical SCLC transcriptional programs, we hypothesized that subtype-specific patterns of DNA methylation could be detected in tumor or blood from SCLC patients. Using genomic-wide reduced-representation bisulfite sequencing (RRBS) in two cohorts totaling 179 SCLC patients and using machine learning approaches, we report a highly accurate DNA methylation-based classifier (SCLC-DMC) that can distinguish SCLC subtypes. We further adjust the classifier for circulating-free DNA (cfDNA) to subtype SCLC from plasma. Using the cfDNA classifier (cfDMC), we demonstrate that SCLC phenotypes can evolve during disease progression, highlighting the need for longitudinal tracking of SCLC during clinical treatment. These data establish that tumor and cfDNA methylation can be used to identify SCLC subtypes and might guide precision SCLC therapy.
(Methods section)
Critical commercial assays
Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit Apostle Bio A17622-250
Nucleic acid extraction
cfDNA was extracted using the Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit (Apostle Inc).
78. Terminal modifications independent cell-free RNA sequencing enables sensitive early cancer detection and classification. Jun Wang, Jinyong Huang, Yunlong Hu, et al. Nature Communications 15, Article number: 156 (2024)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Cell-free RNAs (cfRNAs) offer an opportunity to detect diseases from a transcriptomic perspective, however, existing techniques have fallen short in generating a comprehensive cell-free transcriptome profile. We develop a sensitive library preparation method that is robust down to 100 µl input plasma to analyze cfRNAs independent of their 5’-end modifications. We show that it outperforms adapter ligation-based method in detecting a greater number of cfRNA species. We perform transcriptome-wide characterizations in 165 lung cancer, 30 breast cancer, 37 colorectal cancer, 55 gastric cancer, 15 liver cancer, and 133 cancer-free participants and demonstrate its ability to identify transcriptomic changes occurring in early-stage tumors. We also leverage machine learning analyses on the differentially expressed cfRNA signatures and reveal their robust performance in cancer detection and classification. Our work sets the stage for in-depth study of the cfRNA repertoire and highlights the value of cfRNAs as cancer biomarkers in clinical applications.
Abstract Cell-free RNAs (cfRNAs) offer an opportunity to detect diseases from a transcriptomic perspective, however, existing techniques have fallen short in generating a comprehensive cell-free transcriptome profile. We develop a sensitive library preparation method that is robust down to 100 µl input plasma to analyze cfRNAs independent of their 5’-end modifications. We show that it outperforms adapter ligation-based method in detecting a greater number of cfRNA species. We perform transcriptome-wide characterizations in 165 lung cancer, 30 breast cancer, 37 colorectal cancer, 55 gastric cancer, 15 liver cancer, and 133 cancer-free participants and demonstrate its ability to identify transcriptomic changes occurring in early-stage tumors. We also leverage machine learning analyses on the differentially expressed cfRNA signatures and reveal their robust performance in cancer detection and classification. Our work sets the stage for in-depth study of the cfRNA repertoire and highlights the value of cfRNAs as cancer biomarkers in clinical applications.
(Methods section) cfRNA extraction
Frozen plasma samples were thawed on ice prior to cfRNA extraction. 200 μl of plasma samples were subjected to cfRNA extraction using the Apostle MiniMax High-Efficiency cfRNA Isolation Kit (Apostle), following the manufacturer’s protocol with minor modifications.
77. Extensive methylation analysis of circulating tumor DNA in plasma of patients with gastric cancer. Shinnosuke Nagano, Yukinori Kurokawa, Takaomi Hagi, et al. Scientific Reports 14, Article number: 30739 (2024).
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
DNA methylation is known to be involved in tumor progression. This is the first study to perform an extensive methylation analysis of plasma circulating tumor DNA (ctDNA) using targeted bisulfite sequencing in gastric cancer (GC) patients to evaluate the usefulness of ctDNA methylation as a new biomarker. Sixteen patients who received chemotherapy for recurrent GC were included. After confirmation of the methylation status of 63 genes using the Cancer Genome Atlas (TCGA) dataset, the methylation status in paired tumor and non-tumor tissues and plasma were investigated using targeted bisulfite sequencing in these genes. Forty-four of the 63 genes were significantly hypermethylated in GC patients in the TCGA cohort. Of these 44 genes, hierarchical clustering showed that five (SPG20, FBN1, SDC2, TFPI2, SEPT9) were particularly hypermethylated in tumor compared to non-tumor tissues in our GC cohort. In plasma methylation analysis, patients with high methylation of these genes had significantly worse overall survival than those with low methylation (log-rank P = 0.009). In a patient who underwent blood sampling at multiple points, the methylation levels of these five genes varied closely with clinical tumor status. The plasma ctDNA methylation levels of these five genes could be useful as a noninvasive prognostic biomarker for GC.
(Methods section)
DNA preparation
DNA from plasma samples was isolated using the Apostle MiniMax™ High Efficiency cfDNA Isolation Kit (Beckman Coulter, CA, USA).
76. A novel SNP-based approach for non-invasive prenatal paternity testing using multiplex PCR targeted capture sequencing. Yiling Qu, Ranran Zhang, Li Qing, et al. Journal of Translational Genetics and Genomics. 2024;8:378-93.
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objective: To enhance the safety, simplicity, and efficacy of non-invasive prenatal paternity testing, we developed a method based on multiplex PCR targeted capture sequencing technology utilizing single nucleotide polymorphisms (SNPs) as genetic markers.
Method: We screened 627 SNPs from public databases and literature based on specific criteria and population genetic data from 100 unrelated individuals. A total of 15 peripheral blood samples were collected from pregnant women and the suspected father. Paternal alleles were detected and analyzed in the plasma cell-free DNA (cfDNA) of pregnant women, fetal SNP genotypes were obtained, and the combined paternity index (CPI) was calculated for paternity testing.
Results: Biological fathers were accurately determined in all cases, with CPI values ranging from 1.05 × 1014 to 2.03 × 1034, consistent with results obtained using polymerase chain reaction-capillary electrophoresis (PCR-CE) with short tandem repeats. Significant differences in CPI between unrelated males and biological fathers allowed for straightforward exclusion. Even cfDNA from maternal plasma as early as five gestational weeks enabled accurate paternity determination.
Conclusion: This novel approach demonstrates significant improvements by reducing the number of SNPs, streamlining the research procedure, and lowering costs, yielding substantial advancements in non-invasive prenatal paternity testing.
(Methods and Materials section)
Sample collection
Maternal blood samples (approximately 10 mL) were collected using MiniMax cfDNA blood collection tubes (Apostle, USA)
75. Comprehensive Longitudinal ctDNA Monitoring in Metastatic Cancer Patients Treated with an Individualized Neoantigen-directed Vaccine. Desiree Schenk, Matthew J. Davis, Rita Zhou, Alexis Mantilla, Madeline Galbraith, Oliver Spiro, Olivia Petrillo, Italo Faria do Valle, Andrew R. Ferguson, Karin Jooss, Ankur Dhanik. bioRxiv (2024), https://doi.org/10.1101/2024.12.04.626817
(Note: Apostle MiniMax technology is used in this study.)
Abstract
Purpose Circulating-tumor DNA (ctDNA) is an emerging, minimally invasive diagnostic and prognostic biomarker for patients receiving a variety of cancer therapies. Comprehensive and robust longitudinal monitoring of ctDNA can provide an understanding of tumor burden, heterogeneity, and response or resistance to treatment.
Experimental Design ctDNA of 28 metastatic cancer patients receiving an individualized neoantigen-directed immunotherapy was monitored longitudinally, up to two years, using a unique hybrid next generation sequencing assay targeting tumor-informed and tumor-naïve variants. Patient-specific panels were designed targeting an average of 144 variants per patient. A tumor-naïve universal panel was also designed for inclusion with patient-specific panels to monitor recurrently mutated tumor hotspots (e.g., KRAS and TP53) and genes implicated in immunotherapy resistance (B2M, TAP1/2).
Results Analytical characterization of the assay established linearity with a mean variant allele frequency (VAF) ≥0.049%, and a variant-level limit of detection (LOD95) of 0.12%. Tumor-informed variants were detected in 26/28 patients, and de novo variants were observed in 25/28 patients. HLA LOH was also observed. Longitudinal ctDNA data provided key insights into patients’ responses to vaccine treatment.
Conclusions The hybrid design of the ctDNA monitoring assay provides the sensitivity and specificity required for evaluating patient samples undergoing individualized therapy. It provides an improved capability to understand patient response to experimental therapies and further supports the utility of ctDNA as a cancer biomarker.
(Materials and Methods section)
cfDNA extraction
cfDNA was extracted using the Apostle MiniMax High Efficiency cfDNA Extraction kit (Apostle Bio, San Jose, CA).
74. Liquid Biopsy for Evaluating Mutations and Chromosomal Aberrations in Cerebrospinal Fluid from Patients with Primary or Metastatic Central Tumors. Ahmad Charifa, Sally Agersborg, Arash Mohtashamian, Andrew Ip, Andre Goy, Maher Albitar, The Journal of Liquid Biopsy (2024), https://doi.org/10.1016/j.jlb.2024.100281
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
Cytopathology analysis of cerebrospinal fluid (CSF) is limited in detecting tumors in patients with suspected primary or metastatic central nervous system (CNS) malignancy. We investigated the use of CSF liquid biopsy (LBx) to detect neoplastic processes in the CNS. Methods: Cell-free DNA (cfDNA) from the CSF of patients with suspected metastatic (N=106) or primary CNS (N=23) tumors was deep sequenced using a 302-gene panel.
Results
Four samples (3%) (3 metastatic and 1 primary) failed sequencing quality control criteria. Metastatic tumor was confirmed in 84 (82%) of the 103 patients suspected of metastatic tumor. Primary CNS tumor was confirmed in 11 of 22 (50%) patients suspected of CNS tumor. Chromosomal abnormalities were detected in 55 samples (54%). Germline mutations were detected in 23 (22%) patients with metastatic tumors and in 1 (5%) with a primary CNS tumor. Of the 29 patients with metastatic breast cancers, 2 (7%) had mutations in ESR1 and 9 (31%) had mutations in PIK3CA. Of the 21 patients with metastatic lung cancer, 9 (43%) had EGFR mutations and 5 (24%) had KRAS mutations. Upon comparing CSF LBx with peripheral blood LBx in 14 patients, 13 (93%) showed only CHIP and one patient showed CNS primary tumor mutation. Serial samples from 14 patients demonstrate that CSF LBx can be used for monitoring therapy efficacy.
Conclusions
LBx using CSF is clinically reliable and provides informative results in a substantial proportion of patients with metastatic CNS tumors and to a lesser degree in patients with primary CNS tumors.
(Materials and Methods section)
cfDNA and cfRNA Extraction
We used the Apostle MiniMax High-Efficiency total nucleic acid isolation Kit (Beckman Coulter, Brea, CA, USA) and followed the protocol recommended by the manufacturer as previously described in detail. After extraction, half of the cell-free total nucleic acid was treated with DNase to obtain cfRNA, and the other half was used for cfDNA analysis.
73. CancerSpot: A multi-cancer early detection test developed and validated on a retrospective cohort. Swaraj Basu, Prakash Hiremath M, Nihesh Rathod, et al. medRxiv (2024), doi: https://doi.org/10.1101/2024.12.03.24318395
(Note: Apostle MiniMax technology is used in this study.)
Abstract
Next-generation sequencing (NGS) technologies have transformed biomarker discovery, enabling the detection of disease-associated markers at the earliest stages of illness. In this study, we introduce a blood-based, non-invasive test for multi-cancer detection using cell-free DNA (cfDNA) methylation sequencing. The test employs a novel methylation scoring system derived from sequencing data and integrates machine learning to analyze a retrospective cohort of newly diagnosed cancer cases and controls recruited from multiple centers across India. To enhance robustness, the study includes a substantial proportion of controls with habitual tobacco and alcohol use, ensuring the test’s resilience against confounding factors. The test’s accuracy was further validated through synthetic data augmentation, demonstrating reliability under conditions of random signal perturbation. At an approximate specificity of 97%, the assay achieves sensitivities of 79.3% for Stage I, 78.4% for Stage II, 78.4% for Stage III, and 86.8% for Stage IV cancers in an independent validation cohort. Additionally, the test demonstrates Top 2 Tissue of Origin (TOO) accuracies of 78.3% for Stage I, 79.3% for Stage II, 82.8% for Stage III, and 69.7% for Stage IV cancers. This blood-based test holds considerable promise for early cancer detection, offering a precise test for cancer screening.
(Methods and Materials section)
Methylation assay and sequencing
Cell-free DNA (cfDNA) was extracted from 4 mL of plasma using the Apostle MiniMax High Efficiency cfDNA Isolation Kit (Cat. No.: A17622-384) following the manufacturer’s protocol (Apostle Bio).
72. The pattern of homologous recombination associated genemutations in circulating tumor DNA and solid tumors from pre-treatment ovarian cancer patients reveals potentially usefulpatterns of mutation. John Nakayama, Stephanie Gaillard, Louis Gil Acevedo, et al. Gynecologic Oncology. Volume 190, Supplement 1, S177, November 2024 (Conference abstract)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objectives
This study aimed to assess the ability of comprehensive genomic profiling to identify gene mutations associated with homologous recombination (HR) in the circulating tumor DNA and the solid tumor samples from pre-treatment ovarian cancer patients.
Methods
Samples from 29 ovarian cancer patients were selected from our internal biobank based on the availability of longitudinal samples, as this was the first step of a serial analysis. Comprehensive genomic profiling (CGP) was performed using a validated in-house pan-cancer panel (523 genes), including 25 HR genes, to compare precisely matched solid tumor and blood samples. Pre-treatment blood samples were collected in tubes containing a membrane stabilizer (Streck), and plasma was processed within 72 h to obtain germline-free samples. DNA was extracted from matching formalin-fixed paraffin-embedded tumor tissues and blood using magnetic beads (FormaPure XL, Apostle MiniMax, respectively), and CGP was performed on both samples using the same targeted panel (ct-DNA: NovaSeq6000; st-DNA: NextSeqDx550). The combination of HR mutations found in the solid tumor and blood sample was used in this analysis. Homologous recombination deficiency (HRD+) was defined as either germline or somatic BRCA mutation or positive HRD testing by a commercial vendor. HRD groups were compared using a t-test. A clinically actionable mutation was determined using the OncoKB database.
Results
There were 9 HRD-positive and 10 HRD-negative (all epithelial, stage III & IV) along with 10 HRD unknown patients (5 stage I & II including 1 serous borderline and 1 granulosa cell). Analysis revealed that regardless of stage or histology, every ovarian cancer patient has non-synonymous coding mutations in HR genes. There was a mean of 16.4 (SD: 4.7) coding variants per patient in the total population. A 4-gene (6 variant) signature captured all 29 patients with 18 sharing a BARD1 mutation (c.1518_1519delTGinsCT: Val507Leu) along with FANCA, ATR, and RAD51D single-nucleotide variants. When separated by HRD status, the mean number of mutations per patient was 18.0 (SD: 4.4) in the HRD-positive versus 16.7 (SD: 4.5) in the HRD- negative patients (P = 0.52). Clinically actionable mutations (PARP inhibitors) were identified in 7 of 9 HRD-positive and 4 of 10 HRD-negative patients.
Conclusions
These data show that non-synonymous mutations in HR genes are present in all ovarian cancer patients evaluated. The most common gene variants were found in BARD1, BRCA1, FANCA, ATM, and ATR. A 4-gene mutation signature of ATR, BARD1, FANCA, and RAD51 captured all 29 patients in the study across all stages. Four of 10 HRD-negative patients had clinically actionable mutations, which, if validated in future studies, could open new patients to PARP inhibitor therapy.
71. Comprehensive genomic profiling of circulating tumor DNA ishighly concordant with solid tumor DNA and identifies additionalactionable mutations among advanced ovarian and endometrialcancer patients. John Nakayama, Patricia Petrosko, Phillip Gallo, et al. Gynecologic Oncology. Volume 190, Supplement 1, S167, November 2024 (Conference abstract)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Objectives
This study aimed to determine the accuracy and concordance of circulating tumor DNA (ctDNA) blood samples in providing relevant diagnostic information for patients with advanced ovarian cancer
(OC) and endometrial cancer (EC).
Methods
Advanced OC and EC patients with time-matched, pre-treatment solid tumor and blood samples were identified from our internal biobank. Comprehensive genomic profiling (CGP) was performed using an in-house, validated pan-cancer panel (523 genes). Blood samples were collected using tubes containing a membrane stabilizer (Streck), with plasma processed within 72 hours. DNA was extracted from matching formalin-fixed paraffin-embedded (FFPE) tumor tissues and blood (ctDNA) for each patient using magnetic beads (FormaPure XL, Apostle MiniMax, respectively), and CGP was performed on both samples using the same targeted panel (ctDNA: NovaSeq6000; stDNA: NextSeqDx550). SNV and InDel variants were identified and classified from non-synonymous mutations for clinical actionability using the OncoKB database.
Results
Blood samples from 24 OC and 15 EC patients with stage II-IV disease prior were evaluated. Analysis revealed a mean of 167.2 (SD = 10.0) non-synonymous coding mutations in ovarian solid tumors (ST), with 97.4 % of these mutations (M = 163.0; SD = 10.7) detected in matched ctDNA specimens. Similarly, 181.7 (SD = 21.6) coding mutations were detected in endometrial cancers, with 97.6 % of these found in matched ctDNA specimens (M = 177.6; SD = 21.4). A mean of 19.7 and 14.5 additional mutations/patient in OC and EC samples, respectively, were detected only in the ctDNA as compared to ST specimens. When confined to oncogenic variants defined by OncoKB, 94.3 % (M = 7.0; SD = 2.2) of ovarian and 93.0 % (M = 6.8; SD = 1.8) of endometrial variants in the ST were detected by the ctDNA assay. The ctDNA assay identified an additional 5.0 and 3.1 mutations/patient in the OC and EC samples, respectively. Eleven OC patients were identified to have actionable mutations for targeted antineoplastic therapy using both assays, and an additional 4 patients were identified with actionable targets only in the ctDNA samples. Among EC patients, 7 were identified with actionable mutations in both blood and tumor samples, while only 1 patient had an actionable mutation based on the ctDNA assay alone. Only 1 patient among all 39 patients evaluated had an actionable mutation detected in the ST assay that was absent in the ctDNA assay.
Conclusions
A high concordance exists between paired ctDNA and ST assays from patients with advanced-stage OC and EC. We have demonstrated that ctDNA assays can detect additional actionable mutations not detected in the ST assays. These data provide evidence that ctDNA CGP assays detect additional clinically relevant variants beyond those found in ST assays alone and may be a useful addition to the current standard of ST testing.
70. Liquid Biopsy Assay to Detect Low Levels of ctDNA in Early-Stage Lung Cancer Samples. M. Smith, M. Salmans, M. Wang, et al. Journal of Thoracic Oncology. Volume 19, Issue 10, Supplement, October 2024, Page S474 (Conference abstract)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Introduction
A fundamental challenge in technologies designed to analyze circulating tumor DNA (ctDNA) is the typically small fraction of cell-free DNA (cfDNA) in a blood sample that comes from tumor cells. This proportion of ctDNA is generally low in early-stage cancers, which are characterized by small, localized tumors, but increases as the cancer progresses and the tumors grow and metastasize. Therefore, the capability to reliably detect minimal quantities of ctDNA is crucial for technologies like liquid biopsies. These technologies are particularly focused on detecting cancers at an early stage, when the tumor size is small and the amount of ctDNA present in the bloodstream is minimal. Early detection is especially relevant in lung cancer, where American Cancer Society studies show that the 5-year survival rates plummet from 65% to 9% as the stage at diagnosis advances. Various approaches are being explored to increase the amounts of ctDNA available for analysis, including simply increasing the amount of blood required from the patient paired with ultra-deep targeted sequencing. A low-pass whole genome sequencing approach, in which global cancer-like patterns can be detected in minimal amounts of cfDNA, can provide a reliable indicator of tumor presence without requiring prohibitive amounts of input material. Here we demonstrate the ability of the Genece Health Lung Cancer Assay to leverage fragment and end-motif analysis of cfDNA to detect low levels of ctDNA from early-stage lung cancer samples, while only requiring minimal input material. With the cfDNA available from a single tube of blood from a patient, the Genece Health Lung Cancer Assay can detect all stages of cancer.
Methods
Plasma collected in Streck cfDNA BCT devices from 19 early-stage lung cancer samples (16 stage I samples & 3 stage II samples) were obtained from Discovery Life Sciences. Two plasma aliquots of each sample were extracted using the Apostle MiniMax High Efficiency cfDNA Isolation kit on the KingFisher instrument. Eluates were pooled, cfDNA was quantified using Agilent Tapestation, and then diluted in water to 2ng, 1ng, 0.5ng & 0.25ng, as possible, based on the measured cfDNA concentration.
Results
All early-stage lung cancer samples were detected neat, and subsequent dilution series of these early-stage samples established that an input of only 1ng cfDNA is required for utilization of the Genece Health Lung Assay.
Conclusions
Signal detection in these early-stage lung cancer samples, even after dilution, indicates that the Genece Health Lung Assay has a sufficiently low limit of detection to ensure highly sensitive performance in early-stage lung cancer samples with only a single tube of blood or less.
69. Extracting regulatory active chromatin footprint from cell-free DNA. Kevin Lai, Katharine Dilger, Rachael Cunningham, et al. Communications Biology volume 7, Article number: 1086 (2024)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Cell-free DNA (cfDNA) has emerged as a pivotal player in precision medicine, revolutionizing the diagnostic and therapeutic landscape. While its clinical applications have significantly increased in recent years, current cfDNA assays have limited ability to identify the active transcriptional programs that govern complex disease phenotypes and capture the heterogeneity of the disease. To address these limitations, we have developed a non-invasive platform to enrich and examine the active chromatin fragments (cfDNAac) in peripheral blood. The deconvolution of the cfDNAac signal from traditional nucleosomal chromatin fragments (cfDNAnuc) yields a catalog of features linking these circulating chromatin signals in blood to specific regulatory elements across the genome, including enhancers, promoters, and highly transcribed genes, mirroring the epigenetic data from the ENCODE project. Notably, these cfDNAac counts correlate strongly with RNA polymerase II activity and exhibit distinct expression patterns for known circadian genes. Additionally, cfDNAac signals across gene bodies and promoters show strong correlations with whole blood gene expression levels defined by GTEx. This study illustrates the utility of cfDNAac analysis for investigating epigenomics and gene expression, underscoring its potential for a wide range of clinical applications in precision medicine.
Abstract Cell-free DNA (cfDNA) has emerged as a pivotal player in precision medicine, revolutionizing the diagnostic and therapeutic landscape. While its clinical applications have significantly increased in recent years, current cfDNA assays have limited ability to identify the active transcriptional programs that govern complex disease phenotypes and capture the heterogeneity of the disease. To address these limitations, we have developed a non-invasive platform to enrich and examine the active chromatin fragments (cfDNAac) in peripheral blood. The deconvolution of the cfDNAac signal from traditional nucleosomal chromatin fragments (cfDNAnuc) yields a catalog of features linking these circulating chromatin signals in blood to specific regulatory elements across the genome, including enhancers, promoters, and highly transcribed genes, mirroring the epigenetic data from the ENCODE project. Notably, these cfDNAac counts correlate strongly with RNA polymerase II activity and exhibit distinct expression patterns for known circadian genes. Additionally, cfDNAac signals across gene bodies and promoters show strong correlations with whole blood gene expression levels defined by GTEx. This study illustrates the utility of cfDNAac analysis for investigating epigenomics and gene expression, underscoring its potential for a wide range of clinical applications in precision medicine.
(Results Section, Figure 1) Fig. 1: Active chromatin capture workflow enriches for regulatory-active chromatin fragments in plasma.
a Blood was collected, and plasma separation completed within 24 hours. cfDNA extraction using the off-the-shelf workflow followed the Apostle MiniMax cfDNA protocol to enrich for nucleosomal fragments (cfDNAnuc). The active chromatin capture workflow used optimized extraction and library preparation conditions to enrich for regulatory-active chromatin fragments (cfDNAac), as described in the Methods section. Prepared libraries were then sequenced on a 200 cycle NovaSeq 6000 S4 flowcell. b Size distributions of sequenced cfDNA fragments from subject AS-003, extracted using the off-the-shelf workflow (gray) and the chromatin capture workflow (red).
(Methods Section) cfDNAac extraction and sequencing library preparation
The Apostle kit (Apostle Bio, Pleasanton, CA) has been used in many studies as a standard cfDNA extraction workflow42,43,44,45,46. The Apostle MiniMax High Efficiency cfDNA Isolation Kit was used as the off-the-shelf extraction kit because of its superior performance across a range of input volumes and ability to recover high quantities of cfDNA from different biofluids. Modifications to the Apostle kit were made for our cfDNAac-enriched capture workflow. To optimize for cfDNAac capture, we tested different volumes of lysis (60–200 μL) and binding buffers (800–2000 μL) while maintaining a fixed plasma input volume of 2 mL. Optimal capture of active chromatin cfDNA fragments was achieved with a final volume of 100 μL and 1250 μL for lysis and binding buffers, respectively. The sample lysis step was modified by heating it at 58 °C for 30 min, rather than 60 °C for 20 min.
In both the off-the-shelf and modified workflows, magnetic nanoparticles provided by the commercial kit (Apostle) were used.
68. Evaluation and integration of cell-free DNA signatures for detection of lung cancer. Ruyue Xue, Xiaomin Li, Lu Yang, et al. Cancer Letters Volume 604, 1 November 2024, 217216. https://doi.org/10.1016/j.canlet.2024.217216
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Cell-free DNA (cfDNA) analysis has shown potential in detecting early-stage lung cancer based on non-genetic features. To distinguish patients with lung cancer from healthy individuals, peripheral blood were collected from 926 lung cancer patients and 611 healthy individuals followed by cfDNA extraction. Low-pass whole genome sequencing and targeted methylation sequencing were conducted and various features of cfDNA were evaluated. With our customized algorithm using the most optimal features, the ensemble stacked model was constructed, called ESim-seq (Early Screening tech with Integrated Model). In the independent validation cohort, the ESim-seq model achieved an area under the curve (AUC) of 0.948 (95% CI: 0.915–0.981), with a sensitivity of 79.3% (95% CI: 71.5–87.0%) across all stages at a specificity of 96.0% (95% CI: 90.6–100.0%). Specifically, the sensitivity of the ESim-seq model was 76.5% (95% CI: 67.3–85.8%) in stage I patients, 100% (95% CI: 100.0–100.0%) in stage II patients, 100% (95% CI: 100.0–100.0%) in stage III patients and 87.5% (95% CI: 64.6%–100.0%) in stage IV patients in the independent validation cohort. Besides, we constructed LCSC model (Lung Cancer Subtype multiple Classification), which was able to accurately distinguish patients with small cell lung cancer from those with non-small cell lung cancer, achieving an AUC of 0.961 (95% CI: 0.949–0.957). The present study has established a framework for assessing cfDNA features and demonstrated the benefits of integrating multiple features for early detection of lung cancer.
Abstract Cell-free DNA (cfDNA) analysis has shown potential in detecting early-stage lung cancer based on non-genetic features. To distinguish patients with lung cancer from healthy individuals, peripheral blood were collected from 926 lung cancer patients and 611 healthy individuals followed by cfDNA extraction. Low-pass whole genome sequencing and targeted methylation sequencing were conducted and various features of cfDNA were evaluated. With our customized algorithm using the most optimal features, the ensemble stacked model was constructed, called ESim-seq (Early Screening tech with Integrated Model). In the independent validation cohort, the ESim-seq model achieved an area under the curve (AUC) of 0.948 (95% CI: 0.915–0.981), with a sensitivity of 79.3% (95% CI: 71.5–87.0%) across all stages at a specificity of 96.0% (95% CI: 90.6–100.0%). Specifically, the sensitivity of the ESim-seq model was 76.5% (95% CI: 67.3–85.8%) in stage I patients, 100% (95% CI: 100.0–100.0%) in stage II patients, 100% (95% CI: 100.0–100.0%) in stage III patients and 87.5% (95% CI: 64.6%–100.0%) in stage IV patients in the independent validation cohort. Besides, we constructed LCSC model (Lung Cancer Subtype multiple Classification), which was able to accurately distinguish patients with small cell lung cancer from those with non-small cell lung cancer, achieving an AUC of 0.961 (95% CI: 0.949–0.957). The present study has established a framework for assessing cfDNA features and demonstrated the benefits of integrating multiple features for early detection of lung cancer.
(Methods section) Sample collection and cfDNA extraction
Two 10-mL tubes of peripheral blood from patients were collected using Apostle MiniMax cfDNA Blood Collection Tubes (Apostle) and were gently inverted 10 times to mix immediately. The median time between blood collection and plasma isolation was <2 days (range: 1-5 days). Plasma was separated by two-step centrifugation process.30 Lung cancer and paired para-cancerous tissue samples were collected at the time of surgical resection from the First Affiliated Hospital of Zhengzhou University. Lung cancer tissues with less than 20% tumor cell content were excluded from the study. cfDNA was extracted from 8 to 10 mL of plasma using the Apostle MiniMax High Efficiency cfDNA Isolation Kit (Apostle) on an Apostle MagTouch 2000 Nucleic Acids Extraction Automation System (Apostle), and stored in LoBind tubes (Eppendorf AG). Genomic DNA (gDNA) was extracted from formalin-fixed paraffin-embedded (FFPE) tissue samples using the QIAamp DNA FFPE Tissue Kit (Qiagen) according to the manufacturer’s protocol. The concentration and quality of cfDNA and gDNA were assessed using the Qubit dsDNA HS Assay Kit (Thermo Fisher Scientific) on a Qubit 4 Fluorometer (Thermo Fisher Scientific).
67. Ultra-sensitive molecular residual disease detection through whole genome sequencing with single-read error correction. Xinxing Li, Tao Liu, Antonella Bacchiocchi, et al. EMBO Molecular Medicine (2024) https://doi.org/10.1038/s44321-024-00115-0
(Note: Apostle MiniMax technology is used in this study.)
 Abstract While whole genome sequencing (WGS) of cell-free DNA (cfDNA) holds enormous promise for detection of molecular residual disease (MRD), its performance is limited by WGS error rate. Here we introduce AccuScan, an efficient cfDNA WGS technology that enables genome-wide error correction at single read-level, achieving an error rate of 4.2 × 10−7, which is about two orders of magnitude lower than a read-centric de-noising method. The application of AccuScan to MRD demonstrated analytical sensitivity down to 10−6 circulating variant allele frequency at 99% sample-level specificity. AccuScan showed 90% landmark sensitivity (within 6 weeks after surgery) and 100% specificity for predicting relapse in colorectal cancer. It also showed 67% sensitivity and 100% specificity in esophageal cancer using samples collected within one week after surgery. When AccuScan was applied to monitor immunotherapy in melanoma patients, the circulating tumor DNA (ctDNA) levels and dynamic profiles were consistent with clinical outcomes. Overall, AccuScan provides a highly accurate WGS solution for MRD detection, empowering ctDNA detection at parts per million range without requiring high sample input or personalized reagents.
Abstract While whole genome sequencing (WGS) of cell-free DNA (cfDNA) holds enormous promise for detection of molecular residual disease (MRD), its performance is limited by WGS error rate. Here we introduce AccuScan, an efficient cfDNA WGS technology that enables genome-wide error correction at single read-level, achieving an error rate of 4.2 × 10−7, which is about two orders of magnitude lower than a read-centric de-noising method. The application of AccuScan to MRD demonstrated analytical sensitivity down to 10−6 circulating variant allele frequency at 99% sample-level specificity. AccuScan showed 90% landmark sensitivity (within 6 weeks after surgery) and 100% specificity for predicting relapse in colorectal cancer. It also showed 67% sensitivity and 100% specificity in esophageal cancer using samples collected within one week after surgery. When AccuScan was applied to monitor immunotherapy in melanoma patients, the circulating tumor DNA (ctDNA) levels and dynamic profiles were consistent with clinical outcomes. Overall, AccuScan provides a highly accurate WGS solution for MRD detection, empowering ctDNA detection at parts per million range without requiring high sample input or personalized reagents.
(Methods section) Plasma DNA processing
Isolation of cfDNA from 1 ~ 2 mL of plasma was performed using MiniMax High Efficiency Cell-Free Isolation Kit (catalog no. A17622CN-384, Apostle) and eluted in 80 μL Tris-EDTA buffer.
66. Impact of preanalytical factors on liquid biopsy in the canine cancer model. Kate Megquier, Christopher Husted, Justin Rhoades, et al. bioRxiv July 30, 2024
(Note: Apostle MiniMax technology is used in this study.)
Abstract While liquid biopsy has potential to transform cancer diagnostics through minimally-invasive detection and monitoring of tumors, the impact of preanalytical factors such as the timing and anatomical location of blood draw is not well understood. To address this gap, we leveraged pet dogs with spontaneous cancer as a model system, as their compressed disease timeline facilitates rapid diagnostic benchmarking. Key liquid biopsy metrics from dogs were consistent with existing reports from human patients. The tumor content of samples was higher from venipuncture sites closer to the tumor and from a central vein. Metrics also differed between lymphoma and non-hematopoietic cancers, urging cancer-type-specific interpretation. Liquid biopsy was highly sensitive to disease status, with changes identified soon after post chemotherapy administration, and trends of increased tumor fraction and other metrics observed prior to clinical relapse in dogs with lymphoma or osteosarcoma. These data support the utility of pet dogs with cancer as a relevant system for advancing liquid biopsy platforms.
(Methods section)
Frozen plasma (up to 4mL) was extracted using the Apostle MiniMax cfDNA extraction kit (Apostle, LLC *) on a Biomek i7 Hybrid liquid handling system (Beckman Coulter) per manufacturer instructions.
( * Note from Apostle team: the company name should be Apostle, Inc.)
65. A Blood Hepatocellular Carcinoma Signature Recognizes Very Small Tumor Nodules with Metastatic Traits. Kun Chen, Junxiao Wang, Liping Jiang, et al. Journal of Clinical and Translational Hepatology 2024, doi: 10.14218/JCTH.2023.00559
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background and Aims
Hepatocellular carcinoma (HCC) cases with small nodules are commonly treated with radiofrequency ablation (RFA), but the recurrence rate remains high. This study aimed to establish a blood signature for identifying HCC with metastatic traits pre-RFA.
Methods
Data from HCC patients treated between 2010 and 2017 were retrospectively collected. A blood signature for metastatic HCC was established based on blood levels of alpha-fetoprotein and des-γ-carboxy-prothrombin, cell-free DNA (cfDNA) mutations, and methylation changes in target genes in frozen-stored plasma samples that were collected before RFA performance. The HCC blood signature was validated in patients prospectively enrolled in 2021.
Results
Of 251 HCC patients in the retrospective study, 33.9% experienced recurrence within 1 year post-RFA. The HCC blood signature identified from these patients included des-γ-carboxy-prothrombin ≥40 mAU/mL with cfDNA mutation score, where cfDNA mutations occurred in the genes of TP53, CTNNB1, and TERT promoter. This signature effectively predicted 1-year post-RFA recurrence of HCC with 92% specificity and 91% sensitivity in the retrospective dataset, and with 87% specificity and 76% sensitivity in the prospective dataset (n=32 patients). Among 14 cases in the prospective study with biopsy tissues available, positivity for the HCC blood signature was associated with a higher HCC tissue score and shorter distance between HCC cells and microvasculature.
Conclusions
This study established an HCC blood signature in pre-RFA blood that potentially reflects HCC with metastatic traits and may be valuable for predicting the disease’s early recurrence post-RFA.
(Methods section)
Analysis of cfDNA mutations and methylation changes in stored plasma
Briefly, cfDNA was extracted from the plasma samples using the Apostle MiniMax cfDNA isolation kit. The cfDNA (5–40 ng) was digested using the methylation-sensitive restriction enzyme Hha I (R0139L, New England BioLabs) and subsequently ligated to customized MCP adapters with random DNA barcodes.
64. Optimization of high-volume cell free DNA extraction and end-to-end automation for TSO 500 ctDNA library prep [abstract]. Nripesh Prasad, Rachel Rock, Rebecca Beatty, Melanie Robinson, Dineen Wildman, Michael Sykes, Boris Umylny, Thomas Halsey. In: Proceedings of the American Association for Cancer Research Annual Meeting 2024; Part 1 (Regular Abstracts); 2024 Apr 5-10; San Diego, CA. Philadelphia (PA): AACR; Cancer Res 2024;84(6_Suppl):Abstract nr 2298.
(Note: Apostle MiniMax technology is used and compared in this study.)
 Abstract Liquid biopsy such as plasma, has emerged as one of the most valuable sample types for profiling circulating tumor DNA (ctDNA) for screening, response to treatment, and monitoring relapse in oncology patients. Due to the diffuse nature of ctDNA however, both extraction and library preparation must be optimized for the efficient downstream next-generation sequencing (NGS) application. We initially addressed the optimization of quality and quantity of cfDNA recovery from healthy and various oncology patients by evaluating QIAsymphony DSP Circulating DNA Kit and Apostle MiniMax cfDNA isolation kits. Overall, the cfDNA yields from Apostle MiniMax kit were approximately 12X higher. We then optimized high-volume automated cfDNA extraction from 1ml, 3ml and 6ml plasma samples using the Biomek i7 for Apostle MiniMax kit. Colorectal, Gastric and Ovarian cancer gave the highest yield of cfDNA whereas, Esophageal, Lung and Kidney cancers were the lowest cfDNA yielding samples. We then analyzed ctDNA from plasma from healthy donors, colorectal, kidney, Breast, and endometrial cancer patients along with Oncospan and Seraseq ctDNA mutation mix (5-0.5% AF) to evaluate the mutation detection efficacy, repeatability, and reproducibility by targeted NGS using TSO 500ctDNA assay ay 10ng, 20ng, and 30ng inputs. For library preparation batches of 48 samples were prepared using a fully automated processes for the library prep, Hybridization capture and post capture enrichment on the Beckman i5 robot. Libraries were sequenced on NovaSeq 6000 at 600M Paired-end depth. This method demonstrates an overall concordance of inter-run and intra-run accuracy of variant detection to be 90% demonstrating that this method run as described produces highly repeatable and reproducible variant detection to sub-1% allele frequencies. The inter-run, lot-to-lot, and operator-to-operator variability was ≥99%. Down-sample analysis demonstrated high levels of assay performance with sample inputs of 30 ng with ≥96% sample passing Illumina cut-off metrics at 150M PE read depth as compared to Illumina recommended 420M PE reads. Taken together, we provide a high-volume automated solution for cfDNA extraction from plasma samples, additionally these data demonstrate a robust, reproducible, and highly accurate targeted DNA sequencing method that can be used for low allele frequency variant detection in cfDNA.
Abstract Liquid biopsy such as plasma, has emerged as one of the most valuable sample types for profiling circulating tumor DNA (ctDNA) for screening, response to treatment, and monitoring relapse in oncology patients. Due to the diffuse nature of ctDNA however, both extraction and library preparation must be optimized for the efficient downstream next-generation sequencing (NGS) application. We initially addressed the optimization of quality and quantity of cfDNA recovery from healthy and various oncology patients by evaluating QIAsymphony DSP Circulating DNA Kit and Apostle MiniMax cfDNA isolation kits. Overall, the cfDNA yields from Apostle MiniMax kit were approximately 12X higher. We then optimized high-volume automated cfDNA extraction from 1ml, 3ml and 6ml plasma samples using the Biomek i7 for Apostle MiniMax kit. Colorectal, Gastric and Ovarian cancer gave the highest yield of cfDNA whereas, Esophageal, Lung and Kidney cancers were the lowest cfDNA yielding samples. We then analyzed ctDNA from plasma from healthy donors, colorectal, kidney, Breast, and endometrial cancer patients along with Oncospan and Seraseq ctDNA mutation mix (5-0.5% AF) to evaluate the mutation detection efficacy, repeatability, and reproducibility by targeted NGS using TSO 500ctDNA assay ay 10ng, 20ng, and 30ng inputs. For library preparation batches of 48 samples were prepared using a fully automated processes for the library prep, Hybridization capture and post capture enrichment on the Beckman i5 robot. Libraries were sequenced on NovaSeq 6000 at 600M Paired-end depth. This method demonstrates an overall concordance of inter-run and intra-run accuracy of variant detection to be 90% demonstrating that this method run as described produces highly repeatable and reproducible variant detection to sub-1% allele frequencies. The inter-run, lot-to-lot, and operator-to-operator variability was ≥99%. Down-sample analysis demonstrated high levels of assay performance with sample inputs of 30 ng with ≥96% sample passing Illumina cut-off metrics at 150M PE read depth as compared to Illumina recommended 420M PE reads. Taken together, we provide a high-volume automated solution for cfDNA extraction from plasma samples, additionally these data demonstrate a robust, reproducible, and highly accurate targeted DNA sequencing method that can be used for low allele frequency variant detection in cfDNA.
63. Sequencing of cerebrospinal fluid cell-free DNA facilitated early differential diagnosis of intramedullary spinal cord tumors. Chai, et al. npj Precision Oncology volume 8, Article number: 43 (2024)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Pre-surgery differential diagnosis is valuable for personalized treatment planning in intramedullary spinal cord tumors. This study assessed the performance of sequencing cell-free DNA (cfDNA) in cerebrospinal fluid (CSF) for differential diagnosis of these tumors. Prospectively enrolling 45 patients with intramedullary spinal cord lesions, including diffuse midline glioma (DMG), H3K27-altered (14/45), glioblastoma (1/45), H3-wildtype-astrocytoma (10/45), ependymoma (11/45), and other lesions (9/45), CSF samples were collected via lumbar puncture (41/45), intraoperative extraction (3/45), and Ommaya reservoir (1/45). Then, these samples underwent targeted sequencing along with paired tissue DNA. DMG, H3K27-altered patients exhibited a higher ctDNA positivity (85.7%, 12/14) compared to patients with H3-wildtype-astrocytoma (0/8, P = 0.0003), ependymoma (2/10, P = 0.003), and glioneuronal tumor (0/3, P = 0.009). The histological-grade-IV (P = 0.0027), Ki-67 index ≥10% (P = 0.014), and tumor reaching spinal cord surface (P = 0.012) are also associated with higher ctDNA positivity. Interestingly, for patients with TERT promoter mutant tumors, TERT mutation was detectable in the CSF cfDNA of one DMG case, but not other five cases with histological-grade-II tumors. Shared copy number variants were exclusively observed in DMG, H3K27-altered, and showed a strong correlation (Correlation = 0.95) between CSF and tissue. Finally, H3K27M mutations in CSF exhibited high diagnostic efficiency for DMG, H3K27-altered (Sensitivity = 85.7%, Specificity = 100.0%, AUC = 0.929). Notably, H3K27M was detectable in CSF from patients with recurrent tumors, making it easily applicable for postoperative monitoring. In conclusion, the molecular profile from ctDNA released into CSF of malignant tumors was more frequently detected compared to relatively benign ones. Sequencing of ctDNA in CSF exhibited high efficiency for the differential diagnosis of DMG, H3K27-altered.
(Methods section) Circulating cell-free DNA (cfDNA) isolation from cerebrospinal fluid (CSF)
Then, circulating nucleic acid was extracted from CSF using the Apostle MiniMax High-Efficiency cfDNA Isolation Kit (Apostle, USA) following the manufacturer's instructions.
62. A noninvasive multianalytical approach establishment for risk assessment and gastric cancer screening. Fan, et al. International Journal of Cancer, 2024; 154(6):1111-1123
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Effective screening and early detection are critical to improve the prognosis of gastric cancer (GC). Our study aims to explore noninvasive multianalytical biomarkers and construct integrative models for preliminary risk assessment and GC detection. Whole genomewide methylation marker discovery was conducted with CpG tandems target amplification (CTTA) in cfDNA from large asymptomatic screening participants in a high-risk area of GC. The methylation and mutation candidates were validated simultaneously using one plasma from patients at various gastric lesion stages by multiplex profiling with Mutation Capsule Plus (MCP). Helicobacter pylori specific antibodies were detected with a recomLine assay. Integrated models were constructed and validated by the combination of multianalytical biomarkers. A total of 146 and 120 novel methylation markers were found in CpG islands and promoter regions across the genome with CTTA. The methylation markers together with the candidate mutations were validated with MCP and used to establish a 133-methylation-marker panel for risk assessment of suspicious precancerous lesions and GC cases and a 49-methylation-marker panel as well as a 144-amplicon-mutation panel for GC detection. An integrated model comprising both methylation and specific antibody panels performed better for risk assessment than a traditional model (AUC, 0.83 and 0.63, P < .001). A second model for GC detection integrating methylation and mutation panels also outperformed the traditional model (AUC, 0.82 and 0.68, P = .005). Our study established methylation, mutation and H. pylori-specific antibody panels and constructed two integrated models for risk assessment and GC screening. Our findings provide new insights for a more precise GC screening strategy in the future.
(Methods section) Blood sample collection and DNA extraction
The Apostle MiniMax cfDNA isolation kit (Apostle; San Jose, CA) was used for cfDNA extraction.
61. Dissecting patterns of small cell lung cancer evolution using deep whole genome sequencing of circulating tumor DNA [abstract]. Benjamin B. Morris, Zhihui Zhang, Simon Heeke, et al. In: Proceedings of the AACR Special Conference in Cancer Research: Translating Cancer Evolution and Data Science: The Next Frontier; 2023 Dec 3-6; Boston, Massachusetts. Philadelphia (PA): AACR; Cancer Res 2024;84(3 Suppl_2):Abstract nr A022.
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background: Small cell lung cancer (SCLC) is the most lethal form of lung cancer. A key driver of this near universal lethality is SCLC’s recalcitrance to therapy. The standard of care for SCLC is a chemotherapy doublet of etoposide and cis/carboplatin in combination with immunotherapy (EP+IO). While most tumors respond to these therapies, >80% of SCLCs progress within one year of treatment. Additionally, the aggressive clinical course of SCLC has historically precluded collection of patient-matched treatment naïve and recurrent tumor tissue traditionally required to study evolution. In lieu of tissue, several groups have used circulating tumor DNA (ctDNA) to study SCLC evolution. However, these studies used targeted sequencing panels that analyze <0.04% of the SCLC genome. Using new methods to comprehensively trace how SCLCs rapidly evolve to therapy resistance is a significant unmet need.
Methods: Medical records were reviewed to identify treatment naïve SCLC patients treated at MD Anderson that had both pre- and post-EP+IO liquid biopsies collected under IRB approved protocols. ctDNA was isolated from plasma using a MiniMax High Efficiency Cell-Free DNA Isolation Kit (Apostle Bio). Germline DNA was extracted from buffy coat samples using a QIAamp DNA Blood Midi Kit (Qiagen). Deep whole genome sequencing (WGS) (>100X) of ctDNA and WGS of germline DNA (>60X) was performed using an Illumina NovaSeq X sequencer. Reads were aligned to the hg38 reference genome using BWA-mem. Somatic mutations were called using MuTect2, and clonal and subclonal copy number alterations were identified using Battenberg. DPClust was used to reconstruct cancer cell populations, leveraging mutation and copy number calls.
Results: We identified several deleterious genomic alterations impacting TP53 and RB1 tumor suppressor genes, known genetic drivers of SCLC development. We also detected recurrent, whole chromosome arm loss of heterozygosity events impacting many chromosomes in treatment naïve samples, some of which are known to be present in >80% of SCLCs. Additionally, we found that both treatment naïve and recurrent SCLCs were composed of multiple subclones, each defined by dozens to thousands of unique somatic mutations. COSMIC signature analysis identified high activity of tobacco carcinogens, APOBEC, and replicative-based processes across ctDNA samples. We also identified emergence of novel subclones carrying unique copy number alterations following EP+IO recurrence. Importantly, the affected regions encode key effectors that regulate anti-tumor immune responses and cell survival following DNA damage, both of which are intimately related to the mechanism of action of EP+IO.
Conclusions: Our data shows that deep WGS of ctDNA recapitulates known genetics of SCLC and allows for sensitive characterization of therapy resistant clones. Using this method, we have captured snapshots of both intrinsic and acquired resistance mechanisms to frontline EP+IO. Deep WGS of ctDNA is a promising approach to comprehensively dissect routes of SCLC evolution and therapy resistance.
60. Comprehensive characterization of small noncoding RNA profiles in hypoxia-induced pulmonary hypertension (HPH) rat tissues. Jun Wang, Jiahao Kuang, Shasha Zhang, et al. iScience 2024 Feb 16; 27(2): 108815.
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Hypoxia-induced pulmonary hypertension (HPH) is a fatal cardiovascular disease characterized by an elevation in pulmonary artery pressure, resulting in right ventricular dysfunction and eventual heart failure. Exploring the pathogenesis of HPH is crucial, and small noncoding RNAs (sncRNAs) are gaining recognition as potential regulators of cellular responses to hypoxia. In this study, we conducted a comprehensive analysis of sncRNA profiles in eight tissues of male HPH rats using high-throughput sequencing. Our study unveiled several sncRNAs, with the brain, kidney, and spleen exhibiting the highest abundance of microRNA (miRNA), tRNA-derived small RNA (tDR), and small nucleolar RNA (snoRNA), respectively. Moreover, we identified numerous tissue-specific and hypoxia-responsive sncRNAs, particularly miRNAs and tDRs. Interestingly, we observed arm switching in miRNAs under hypoxic conditions and a significant increase in the abundance of 5′ tRNA-halves among the total tDRs during hypoxia. Overall, our study provides a comprehensive characterization of the sncRNA profiles in HPH rats.
Methods Section
sncRNA sequencing library preparation
To isolate total RNA from the eight different tissues and plasma, we used RNAiso Plus (Takara, Japan) and Apostle MiniMax High Efficiency cfRNA Isolation Kit (Apostle) according to the manufacturer’s instructions.
59. Integration of Cell-Free DNA End Motifs and Fragment Lengths Can Identify Active Genes in Liquid Biopsies. Christoffer Trier Maansson, Louise Skov Thomsen, Peter Meldgaard, et al. International Journal of Molecular Sciences. 2024, 25(2), 1243
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Multiple studies have shown that cell-free DNA (cfDNA) from cancer patients differ in both fragment length and fragment end motif (FEM) from healthy individuals, yet there is a lack of understanding of how the two factors combined are associated with cancer and gene transcription. In this study, we conducted cfDNA fragmentomics evaluations using plasma from lung cancer patients (n = 12) and healthy individuals (n = 7). A personal gene expression profile was established from plasma using H3K36me3 cell-free chromatin immunoprecipitation sequencing (cfChIP-seq). The genes with the highest expression displayed an enrichment of short cfDNA fragments (median = 19.99%, IQR: 16.94–27.13%, p < 0.0001) compared to the genes with low expression. Furthermore, distinct GC-rich FEMs were enriched after cfChIP. Combining the frequency of short cfDNA fragments with the presence of distinct FEMs resulted in an even further enrichment of the most expressed genes (median = 37.85%, IQR: 30.10–39.49%, p < 0.0001). An in vitro size selection of <150 bp cfDNA could isolate cfDNA representing active genes and the size-selection enrichment correlated with the cfChIP-seq enrichment (Spearman r range: 0.499–0.882, p < 0.0001). This study expands the knowledge regarding cfDNA fragmentomics and sheds new light on how gene activity is associated with both cfDNA fragment lengths and distinct FEMs.
Abstract Multiple studies have shown that cell-free DNA (cfDNA) from cancer patients differ in both fragment length and fragment end motif (FEM) from healthy individuals, yet there is a lack of understanding of how the two factors combined are associated with cancer and gene transcription. In this study, we conducted cfDNA fragmentomics evaluations using plasma from lung cancer patients (n = 12) and healthy individuals (n = 7). A personal gene expression profile was established from plasma using H3K36me3 cell-free chromatin immunoprecipitation sequencing (cfChIP-seq). The genes with the highest expression displayed an enrichment of short cfDNA fragments (median = 19.99%, IQR: 16.94–27.13%, p < 0.0001) compared to the genes with low expression. Furthermore, distinct GC-rich FEMs were enriched after cfChIP. Combining the frequency of short cfDNA fragments with the presence of distinct FEMs resulted in an even further enrichment of the most expressed genes (median = 37.85%, IQR: 30.10–39.49%, p < 0.0001). An in vitro size selection of <150 bp cfDNA could isolate cfDNA representing active genes and the size-selection enrichment correlated with the cfChIP-seq enrichment (Spearman r range: 0.499–0.882, p < 0.0001). This study expands the knowledge regarding cfDNA fragmentomics and sheds new light on how gene activity is associated with both cfDNA fragment lengths and distinct FEMs.
(Methods section) Cell-Free Chromatin Immunoprecipitaiton (cfChIP) Enrichment
The cfChIP sample and the input plasma cfDNA were purified using an Apostle MiniMax High Efficiency cfDNA Isolation Kit (Beckman Coulter, Indianapolis, IN, USA) and subjected to CAPP-seq.
Publications (2023)
58. Integrative modeling of tumor genomes and epigenomes for enhanced cancer diagnosis by cell-free DNA. Mingyun Bae, Gyuhee Kim, Tae-Rim Lee, et al. Nature Communications 14, Article number: 2017 (2023)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Multi-cancer early detection remains a key challenge in cell-free DNA (cfDNA)-based liquid biopsy. Here, we perform cfDNA whole-genome sequencing to generate two test datasets covering 2125 patient samples of 9 cancer types and 1241 normal control samples, and also a reference dataset for background variant filtering based on 20,529 low-depth healthy samples. An external cfDNA dataset consisting of 208 cancer and 214 normal control samples is used for additional evaluation. Accuracy for cancer detection and tissue-of-origin localization is achieved using our algorithm, which incorporates cancer type-specific profiles of mutation distribution and chromatin organization in tumor tissues as model references. Our integrative model detects early-stage cancers, including those of pancreatic origin, with high sensitivity that is comparable to that of late-stage detection. Model interpretation reveals the contribution of cancer type-specific genomic and epigenomic features. Our methodologies may lay the groundwork for accurate cfDNA-based cancer diagnosis, especially at early stages.
Abstract Multi-cancer early detection remains a key challenge in cell-free DNA (cfDNA)-based liquid biopsy. Here, we perform cfDNA whole-genome sequencing to generate two test datasets covering 2125 patient samples of 9 cancer types and 1241 normal control samples, and also a reference dataset for background variant filtering based on 20,529 low-depth healthy samples. An external cfDNA dataset consisting of 208 cancer and 214 normal control samples is used for additional evaluation. Accuracy for cancer detection and tissue-of-origin localization is achieved using our algorithm, which incorporates cancer type-specific profiles of mutation distribution and chromatin organization in tumor tissues as model references. Our integrative model detects early-stage cancers, including those of pancreatic origin, with high sensitivity that is comparable to that of late-stage detection. Model interpretation reveals the contribution of cancer type-specific genomic and epigenomic features. Our methodologies may lay the groundwork for accurate cfDNA-based cancer diagnosis, especially at early stages.
(Methods section) cfDNA was extracted from 0.4 mL plasma ... and eluted in a final volume of 22 μL, using an Apostle MiniMax High Efficiency cfDNA Isolation Kit (Apostle, US) according to the manufacturer’s instructions.
57. A CRISPR/Cas12a Based Sensitive and Rapid System for Detecting Hepatitis a Virus in Foods. Li, et al. Heliyon, December 14, 2023; http://dx.doi.org/10.2139/ssrn.4656007
(Note: Apostle MiniGenomics technology is used in this study.)
Abstract
As food can easily become the carrier and spreader of infectious pathogens, food safety has been a global concern because of frequent international food trade. Among the foodborne infectious pathogens, hepatitis A virus(HAV) is one of the most common ones. However, its detection usually requires sophisticated laboratory techniques such as reverse transcription quantitative PCR (RT-qPCR), which prevents it from being widely used as an on-site detection tool. Here, we established a new HAV detection system to lower the requirement for temperature control by combining optimized and simplified CRISPR (clustered regularly interspaced short palindromic repeats) Cas12a_based HOLMES with enhanced-RT-LAMP (reverse transcription loop-mediated isothermal amplification) method. The obtained system, namely HOLMES(HAV), requires only two isothermal temperatures, and the whole procedure can be finished within 40min with the detection sensitivity of 0.4fM. Overall, this study develops an innovative analytical approach for rapid detection of HAV in food samples, which will efficiently identify pathogen-contaminated food and prevent possible human infection.
(Methods section)
RNA was extracted using Viral Total RNA Isolation Fast Kit (Apostle MiniGenomics, CA, USA) according to the manufacturer’s instructions, with 50 μL RNA solution obtained for each sample, which was stored at -80 °C until use.
56. Comprehensive Analysis of Cell-Free DNA Fragmentation Across Cancer Stages. Guo, et al. medRxiv, November 2023; https://doi.org/10.1101/2023.11.07.23298181
(Note: Apostle MiniMax technology is used in this study.)
Abstract
Circulating cell-free DNA (cfDNA) in the bloodstream displays cancer-derived fragmentation patterns, offering a non-invasive diagnostic avenue for cancer patients. However, the association between cfDNA fragmentation patterns and cancer progression remains largely unexplored. In this study, we analyzed this relationship using 214 whole-genome cfDNA samples across seven solid cancer types and revealed that the relation between cfDNA fragmentation patterns and cancer stages vary across cancer types. Among them, cfDNA fragmentation patterns in colorectal cancer (CRC) showed a strong correlation with cancer stages. This finding is further validated with an independent targeted cfDNA dataset from 29 CRC samples. Inspired by these findings, we designed “frag2stage”, a machine learning model that exploits cfDNA fragmentation data to differentiate cancer stages of CRC. Evaluated on two independent cfDNA datasets, our model can distinguish cancer stages of CRC with the area under the curve (AUC) values ranging from 0.68 to 0.99. The results suggest that cfDNA fragmentation patterns might carry yet undiscovered genetic and epigenetic signals, highlighting their promising potential for broader diagnostic applications in oncology.
(Methods section) CFDNA EXTRACTION AND NGS LIBRARY PREPARATION
Blood samples, about 10 ml from each individual, were collected using the Apostle MiniMax cf-DNA Blood Collection Tube (Apostle; San Jose, CA, USA) and processed within seven days from the collection. The cfDNA extraction from 2 to 5 ml of each plasma sample was conducted using the Apostle MiniMax High Efficiency cfDNA isolation kit (Apostle; San Jose, CA, USA), with adherence to the manufacturer’s protocol with slight modifications.
55. Concordance analysis of cerebrospinal fluid with the tumor tissue for integrated diagnosis in gliomas based on next-generation sequencing. Wang, et al. Pathology and Oncology Research, 26 September 2023 https://doi.org/10.3389/pore.2023.1611391
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Purpose: The driver mutations of gliomas have been identified in cerebrospinal fluid (CSF). Here we compared the concordance between CSF and tumor tissue for integrated diagnosis in gliomas using next-generation sequencing (NGS) to evaluate the feasibility of CSF detection in gliomas.
Patients and methods: 27 paired CSF/tumor tissues of glioma patients were sequenced by a customized gene panel based on NGS. All CSF samples were collected through lumbar puncture before surgery. Integrated diagnosis was made by analysis of histology and tumor DNA molecular pathology according to the 2021 WHO classification of the central nervous system tumors.
Results: A total of 24 patients had detectable circulating tumor DNA (ctDNA) and 22 had at least one somatic mutation or chromosome alteration in CSF. The ctDNA levels varied significantly across different ages, Ki-67 index, magnetic resonance imaging signal and glioma subtypes (p < 0.05). The concordance between integrated ctDNA diagnosis and the final diagnosis came up to 91.6% (Kappa, 0.800). We reclassified the clinical diagnosis of 3 patients based on the results of CSF ctDNA sequencing, and 4 patients were reassessed depending on tumor DNA. Interestingly, a rare IDH1 R132C was identified in CSF ctDNA, but not in the corresponding tumor sample.
Conclusion: This study demonstrates a high concordance between integrated ctDNA diagnosis and the final diagnosis of gliomas, highlighting the practicability of NGS based detection of mutations of CSF in assisting integrated diagnosis of gliomas, especially glioblastoma.
(Methods section) DNA extraction and quantification
Cell-free DNA (cfDNA) was extracted using an Apostle MiniMax High Efficiency cfDNA Isolation Kit (APOSTLE) according to the manufacturer’s instructions.
54. Altered cfDNA fragmentation profile in hypomethylated regions as diagnostic markers in breast cancer. Wang J, et al. Epigenetics & Chromatin. 16, Article number: 33 (2023)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Background
Breast cancer, the most common malignancy in women worldwide, has been proven to have both altered plasma cell-free DNA (cfDNA) methylation and fragmentation profiles. Nevertheless, simultaneously detecting both of them for breast cancer diagnosis has never been reported. Moreover, although fragmentation pattern of cfDNA is determined by nuclease digestion of chromatin, structure of which may be affected by DNA methylation, whether cfDNA methylation and fragmentation are biologically related or not still remains unclear.
Methods
Improved cfMeDIP-seq were utilized to characterize both cfDNA methylation and fragmentation profiles in 49 plasma samples from both healthy individuals and patients with breast cancer. The feasibility of using cfDNA fragmentation profile in hypo- and hypermethylated regions as diagnostic markers for breast cancer was evaluated.
Results
Mean size of cfDNA fragments (100–220 bp) mapped to hypomethylated regions decreased more in patients with breast cancer (4.60 bp, 172.33 to 167.73 bp) than in healthy individuals (2.87 bp, 174.54 to 171.67 bp). Furthermore, proportion of short cfDNA fragments (100–150 bp) in hypomethylated regions when compared with it in hypermethylated regions was found to increase more in patients with breast cancer in two independent discovery cohort. The feasibility of using abnormality of short cfDNA fragments ratio in hypomethylated genomic regions for breast cancer diagnosis in validation cohort was evaluated. 7 out of 11 patients were detected as having breast cancer (63.6% sensitivity), whereas no healthy individuals were mis-detected (100% specificity).
Conclusion
We identified enriched short cfDNA fragments after 5mC-immunoprecipitation (IP) in patients with breast cancer, and demonstrated the enriched short cfDNA fragments might originated from hypomethylated genomic regions. Furthermore, we proved the feasibility of using differentially methylated regions (DMRs)-dependent cfDNA fragmentation profile for breast cancer diagnosis.
(Methods section) Sample collection and cfDNA extraction
cfDNA was extracted from plasma using MiniMax(TM) High Efficiency Cell-Free DNA Isolation Kit (Apostle, A17622-250) according to manufacturer’s instructions.
53. Combined detection of SDC2/ADHFE1/PPP2R5C methylation in stool DNA for colorectal cancer screening. Li B, et al. Journal of Cancer Research and Clinical Oncology. June 03, 2023. https://doi.org/10.1007/s00432-023-04943-4
(Note: Apostle MiniGenomics technology is used in this study.)
 Abstract
Abstract
Background Colorectal cancer (CRC) is a disease of global concern, and its increasing incidence suggests the need for early and accurate diagnosis. The aim of this study was to investigate the value of combined detection of SDC2, ADHFE1 and PPP2R5C gene methylation in stool samples for early CRC screening.
Methods StoolsamplesfrompatientswithCRC(n=105),advancedadenoma(AA)(n=54),non-advancedadenoma(NA) (n = 57), hyperplastic or other polyps (HOP) (n = 47) or no evidence of disease (NED) (n = 100) were collected from Sep- tember 2021 to September 2022. The methylation levels of SDC2, ADHFE1 and PPP2R5C were quantified by quantitative methylation-specific polymerase chain reaction (qMSP), and faecal immunochemical testing (FIT) was performed. The diagnostic value was assessed using reporter operating characteristic (ROC) curve analysis.
Results The sensitivity of combined detection of SDC2/ADHFE1/PPP2R5C methylation in predicting CRC (0–IV) was 84.8%, the specificity was 98.0%, and the AUC was 0.930 (95% CI 0.889–0.970). Compared to FIT and serum tumour bio- markers, it showed better diagnostic performance for different stages of CRC.
Conclusion The results of this study verified that the methylation levels of SDC2, ADHFE1 and PPP2R5C in stool DNA were significantly increased in CRC patients. Combined detection of SDC2/ADHFE1/PPP2R5C methylation is a potential non-invasive diagnostic method for CRC and precancerous lesion screening.
Clinical trial registration Chinese Clinical Trials Registry, ChiCTR2100046662, registered on 26 May 2021, prospective registration.
(Methods section) DNA isolation, bisulphite treatment and fluorescence‐based quantitative methylation‐specific polymerase chain reaction (qMSP)
Genomic DNA was isolated from stool samples using the Apostle MiniGenomics (TM) Stool Fast Kit (Apostle, A181206) and stored in the refrigerator at – 20 °C until further use.
52. Combining cell-free RNA with cell-free DNA in liquid biopsy for hematologic and solid tumors. Maher Albitar, Hong Zhang, Ahmad Charifa, et al. Heliyon 9 (2023) e16261; May 16, 2023
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Current use of liquid biopsy is based on cell-free DNA (cfDNA) and the evaluation of mutations or methylation pattern. However, expressed RNA can capture mutations, changes in expression levels due to methylation, and provide information on cell of origin, growth, and proliferation status. We developed an approach to isolate cell-free total nucleic acid (cfDNA) and used targeted next generation sequencing to sequence cell-free RNA (cfRNA) and cfDNA as new approach in liquid biopsy. We demonstrate that cfRNA is overall more sensitive than cfDNA in detecting mutations. We show that cfRNA is reliable in detecting fusion genes and cfDNA is reliable in detecting chromosomal gains and losses. cfRNA levels of various solid tumor biomarkers were significantly higher (P < 0.0001) in samples from solid tumors as compared with normal control. Similarly, cfRNA lymphoid markers and cfRNA myeloid markers were all higher in lymphoid and myeloid neoplasms, respectively as compared with control (P < 0.0001). Using machine learning we demonstrate cfRNA was highly predictive of diagnosis (AUC >0.98) of solid tumors, B-cell lymphoid neoplasms, T-cell lymphoid neoplasms, and myeloid neoplasms. In evaluating the host immune system, cfRNA CD4:CD8B and CD3D:CD19 ratios in normal controls were as expected (median: 5.92 and 6.87, respectively) and were significantly lower in solid tumors (P < 0.0002). This data suggests that liquid biopsy combining analysis of cfRNA with cfDNA is practical and may provide helpful information in predicting genomic abnormalities, diagnosis of neoplasms and evaluating both the tumor biology and the host response.
Abstract Current use of liquid biopsy is based on cell-free DNA (cfDNA) and the evaluation of mutations or methylation pattern. However, expressed RNA can capture mutations, changes in expression levels due to methylation, and provide information on cell of origin, growth, and proliferation status. We developed an approach to isolate cell-free total nucleic acid (cfDNA) and used targeted next generation sequencing to sequence cell-free RNA (cfRNA) and cfDNA as new approach in liquid biopsy. We demonstrate that cfRNA is overall more sensitive than cfDNA in detecting mutations. We show that cfRNA is reliable in detecting fusion genes and cfDNA is reliable in detecting chromosomal gains and losses. cfRNA levels of various solid tumor biomarkers were significantly higher (P < 0.0001) in samples from solid tumors as compared with normal control. Similarly, cfRNA lymphoid markers and cfRNA myeloid markers were all higher in lymphoid and myeloid neoplasms, respectively as compared with control (P < 0.0001). Using machine learning we demonstrate cfRNA was highly predictive of diagnosis (AUC >0.98) of solid tumors, B-cell lymphoid neoplasms, T-cell lymphoid neoplasms, and myeloid neoplasms. In evaluating the host immune system, cfRNA CD4:CD8B and CD3D:CD19 ratios in normal controls were as expected (median: 5.92 and 6.87, respectively) and were significantly lower in solid tumors (P < 0.0002). This data suggests that liquid biopsy combining analysis of cfRNA with cfDNA is practical and may provide helpful information in predicting genomic abnormalities, diagnosis of neoplasms and evaluating both the tumor biology and the host response.
(Methods section) We used Apostle MiniMax High Efficiency cfRNA/cfDNA isolation kit and followed the recommended protocol. After extraction, half of the cfDNA was treated with DNase to obtain cfRNA and the other half was used for DNA studies.
51. Noninvasive Prenatal Screening for Common Fetal Aneuploidies Using Single-Molecule Sequencing. Yeqing Qian, Yongfeng Liu, Kai Yan, et al. Laboratory Investigation Volume 103, Issue 4, April 2023, 100043
(Note: Apostle MiniMax technology is used in this study.)
 Amplification biases caused by next-generation sequencing (NGS) for noninvasive prenatal screening (NIPS) may be reduced using single-molecule sequencing (SMS), during which PCR is omitted. Therefore, the performance of SMS-based NIPS was evaluated. We used SMS-based NIPS to screen for common fetal aneuploidies in 477 pregnant women. The sensitivity, specificity, positive predictive value, and negative predictive value were estimated. The GC-induced bias was compared between the SMS- and NGS-based NIPS methods. Notably, a sensitivity of 100% was achieved for fetal trisomy 13 (T13), trisomy 18 (T18), and trisomy 21 (T21). The positive predictive value was 46.15% for T13, 96.77% for T18, and 99.07% for T21. The overall specificity was 100% (334/334). Compared with NGS, SMS (without PCR) had less GC bias, a better distinction between T21 or T18 and euploidies, and better diagnostic performance. Overall, our results suggest that SMS improves the performance of NIPS for common fetal aneuploidies by reducing the GC bias introduced during library preparation and sequencing.
Amplification biases caused by next-generation sequencing (NGS) for noninvasive prenatal screening (NIPS) may be reduced using single-molecule sequencing (SMS), during which PCR is omitted. Therefore, the performance of SMS-based NIPS was evaluated. We used SMS-based NIPS to screen for common fetal aneuploidies in 477 pregnant women. The sensitivity, specificity, positive predictive value, and negative predictive value were estimated. The GC-induced bias was compared between the SMS- and NGS-based NIPS methods. Notably, a sensitivity of 100% was achieved for fetal trisomy 13 (T13), trisomy 18 (T18), and trisomy 21 (T21). The positive predictive value was 46.15% for T13, 96.77% for T18, and 99.07% for T21. The overall specificity was 100% (334/334). Compared with NGS, SMS (without PCR) had less GC bias, a better distinction between T21 or T18 and euploidies, and better diagnostic performance. Overall, our results suggest that SMS improves the performance of NIPS for common fetal aneuploidies by reducing the GC bias introduced during library preparation and sequencing.
(Materials and Methods section) - cfDNA was extracted from 0.6 mL of maternal plasma using Apostle MiniMaxTM High-Efficiency cfDNA Isolation Kit (ref. number: A17622-50; Apostle) according to the manufacturer’s instructions.
50. Rapid Lineage Assignment of Severe Acute Respiratory Syndrome Coronavirus 2 Cases through Automated Library Preparation, Sequencing, and Bioinformatic Analysis. Andrew J. Gorzalski, Heather Kerwin, Subhash Verma, David C. Hess, Joel Sevinsky, Kevin Libuit, Irina Vlasova-St. Louis, Danielle Siao, Lauren Siao, Diego Buñuel, Stephanie Van Hooser, Mark W. Pandori. The Journal of Molecular Diagnostics Volume 25, Issue 4, April 2023, Pages 191-196
(Note: Apostle MiniGenomics, Apostle MagTouch technologies are used in this study.)
 The coronavirus disease 2019 (COVID-19) pandemic has provided a stage to illustrate that there is considerable value in obtaining rapid, whole-genome–based information about pathogens. This article describes the utility of a commercially available, automated severe acute respiratory syndrome associated coronavirus 2 (SARS-CoV-2) library preparation, genome sequencing, and a bioinformatics analysis pipeline to provide rapid, near–real-time SARS-CoV-2 variant description. This study evaluated the turnaround time, accuracy, and other quality-related parameters obtained from commercially available automated sequencing instrumentation, from analysis of continuous clinical samples obtained from January 1, 2021, to October 6, 2021. This analysis included a base-by-base assessment of sequencing accuracy at every position in the SARS-CoV-2 chromosome using two commercially available methods. Mean turnaround time, from the receipt of a specimen for SARS-CoV-2 testing to the availability of the results, with lineage assignment, was <3 days. Accuracy of sequencing by one method was 100%, although certain sites on the genome were found repeatedly to have been sequenced with varying degrees of read error rate.
The coronavirus disease 2019 (COVID-19) pandemic has provided a stage to illustrate that there is considerable value in obtaining rapid, whole-genome–based information about pathogens. This article describes the utility of a commercially available, automated severe acute respiratory syndrome associated coronavirus 2 (SARS-CoV-2) library preparation, genome sequencing, and a bioinformatics analysis pipeline to provide rapid, near–real-time SARS-CoV-2 variant description. This study evaluated the turnaround time, accuracy, and other quality-related parameters obtained from commercially available automated sequencing instrumentation, from analysis of continuous clinical samples obtained from January 1, 2021, to October 6, 2021. This analysis included a base-by-base assessment of sequencing accuracy at every position in the SARS-CoV-2 chromosome using two commercially available methods. Mean turnaround time, from the receipt of a specimen for SARS-CoV-2 testing to the availability of the results, with lineage assignment, was <3 days. Accuracy of sequencing by one method was 100%, although certain sites on the genome were found repeatedly to have been sequenced with varying degrees of read error rate.
(Material and Methods section) - Nucleic acid was extracted using the MagTouch Nucleic Acid Extraction Automation System (Apostle Inc., San Jose, CA).
49. Cell-free chromatin immunoprecipitation can determine tumor gene expression in lung cancer patients. Christoffer Trier Maansson, Peter Meldgaard, Magnus Stougaard, Anders Lade Nielsen, Boe Sandahl Sorensen. Molecular Oncology. February 24, 2023. DOI: 10.1002/1878-0261.13394.
(Note: Apostle MiniMax technology is used in this study.)
 Cell-free DNA (cfDNA) in blood plasma can be bound to nucleosomes that contain post-translational modifications representing the epigenetic profile of the cell of origin. This includes histone H3 lysine 36 trimethylation (H3K36me3), a marker of active transcription. We hypothesized that cell-free chromatin immunoprecipitation (cfChIP) of H3K36me3-modified nucleosomes present in blood plasma can delineate tumor gene expression levels. H3K36me3 cfChIP followed by targeted NGS (cfChIP-seq) was performed on blood plasma samples from non-small cell lung cancer patients (NSCLC, n = 8), small cell lung cancer patients (SCLC, n = 4) and healthy controls (n = 4). H3K36me3 cfChIP-seq demonstrated increased enrichment of mutated alleles compared to normal alleles in plasma from patients with known somatic cancer mutations. Additionally, genes identified to be differentially expressed in SCLC and NSCLC tumors had concordant H3K36me3 cfChIP enrichment profiles in NSCLC (sensitivity = 0.80) and SCLC blood plasma (sensitivity = 0.86). Findings here expand the utility of cfDNA in liquid biopsies to characterize treatment resistance, cancer subtyping, and disease progression.
Cell-free DNA (cfDNA) in blood plasma can be bound to nucleosomes that contain post-translational modifications representing the epigenetic profile of the cell of origin. This includes histone H3 lysine 36 trimethylation (H3K36me3), a marker of active transcription. We hypothesized that cell-free chromatin immunoprecipitation (cfChIP) of H3K36me3-modified nucleosomes present in blood plasma can delineate tumor gene expression levels. H3K36me3 cfChIP followed by targeted NGS (cfChIP-seq) was performed on blood plasma samples from non-small cell lung cancer patients (NSCLC, n = 8), small cell lung cancer patients (SCLC, n = 4) and healthy controls (n = 4). H3K36me3 cfChIP-seq demonstrated increased enrichment of mutated alleles compared to normal alleles in plasma from patients with known somatic cancer mutations. Additionally, genes identified to be differentially expressed in SCLC and NSCLC tumors had concordant H3K36me3 cfChIP enrichment profiles in NSCLC (sensitivity = 0.80) and SCLC blood plasma (sensitivity = 0.86). Findings here expand the utility of cfDNA in liquid biopsies to characterize treatment resistance, cancer subtyping, and disease progression.
(Material and Methods section) -Both the input as well as the cfChIP samples were purified using Apostle MiniMax High Efficiency cfDNA Isolation Kit (Beckman Coulter, Indianapolis, IN, USA) according to manufacturer’s instructions.
48. Automatic Separation and Collection of Cancer-Related Substances from Clinical Samples. Jin-Han Bae, Jay Jeong, Byung Chul Kim, et al. Journal of Visualized Experiments. January 13th, 2023. DOI: 10.3791/64325
(Note: Apostle MiniMax technology is used in this study.)
Recently, liquid biopsies have been used to diagnose various diseases, including cancer. Body fluids contain many substances, including cells, proteins, and nucleic acids originating from normal tissues, but some of these substances also originate from the diseased area. The investigation and analysis of these substances in the body fluids play a pivotal role in the diagnosis of various diseases. Therefore, it is important to accurately separate the required substances, and several techniques are developed to be used for this purpose. We have developed a lab-on-a-disc type of device and platform named CD-PRIME. This device is automated and has good results for sample contamination and sample stability. Moreover, it has advantages of a good acquisition yield, a short operation time, and high reproducibility. In addition, depending on the type of disc to be mounted, plasma containing cell-free DNA, circulating tumor cells, peripheral blood mononuclear cells, or buffy coats can be separated. Thus, the acquisition of a variety of materials present in the body fluids can be done for a variety of downstream applications, including the study of omics.
(Materials section) - Apostle MiniMax High Efficiency Cell-Free DNA Isolation Kit Apostle A17622-250 5 mL X 50 preps version
47. Comparison of cfDNA content in different cancer types with different extraction methods. Wenlong Zhang, Yanan Zhao, Jun Li, et al. Journal of Clinical Oncology 41, no. 16_suppl (June 01, 2023) e15026-e15026.
(Note: Apostle MiniMax technology is used in this study.)
 Background: ctDNA liquid biopsy technology has been widely used in the whole course of cancer management, from early screening and early diagnosis of tumors to tumor monitoring and medication guidance. However, the performances of ctDNA as a biomarker in these scenarios are different among different cancer types. One possible reason is that different types of cancer release different amounts of ctDNA or cfDNA, therefore, we explored the cfDNA levels in different cancer types in this study.
Background: ctDNA liquid biopsy technology has been widely used in the whole course of cancer management, from early screening and early diagnosis of tumors to tumor monitoring and medication guidance. However, the performances of ctDNA as a biomarker in these scenarios are different among different cancer types. One possible reason is that different types of cancer release different amounts of ctDNA or cfDNA, therefore, we explored the cfDNA levels in different cancer types in this study.
Methods: We collected 15913 peripheral blood samples from 14686 cancer patients with different clinicopathologies, 256 patients with pulmonary nodules, and 971 healthy people. We isolated their plasma and extracted the cfDNA using QIAamp Circulating Nucleic Acid Kit, Apostle MiniMax cfDNA Extraction Kit or HaploX magnetic bead method cfDNA extraction kit. We calculated the average concentration of cfDNA (ng/ml) for different groups.
Results: The number of samples of cervical cancer, liver cancer, glioma, colon cancer, prostate cancer, nasopharyngeal carcinoma, gallbladder carcinoma, melanoma, cholangiocarcinoma, colorectal cancer, breast cancer, gastric cancer, endometrial carcinoma, lung cancer, urothelial carcinoma, rectal cancer, ovarian cancer, healthy people, and pulmonary nodule patients are 93, 469, 113, 277, 139, 76, 89, 132, 171, 4010, 407, 547, 68, 7750, 46, 203, 96, 971 and 256, respectively. The cfDNA level of corresponding groups are 88.07, 79.51, 75.57, 63.58, 61.37, 59.09, 57.59, 57.16, 42.67, 34.73, 31.75, 31.37, 31.33, 29.84, 28.72, 28.69, 23.83, 17.82 and 12.54 ng/ml, respectively. The main groups are shown in the table below.
Conclusions: The cfDNA concentrations of healthy people or pulmonary nodule patients were significantly lower than that of tumor patients, and there were also significant differences between different types of cancer. What's interesting, the cfDNA concentrations of lung cancer patients, pulmonary nodule patients and healthy people were quite different. What’s more, there were differences between Colon cancer and Rectal cancer patients. So, the cfDNA concentration maybe a good dimention to be used in early cancer screening and tracing of tumor types.
Publications (2022)
46. Individualized, heterologous chimpanzee adenovirus and self-amplifying mRNA neoantigen vaccine for advanced metastatic solid tumors: phase 1 trial interim results. Christine D. Palmer, Amy R. Rappaport, Matthew J. Davis, et al. Nature Medicine volume 28, pages 1619–1629 (2022)
(Note: Apostle MiniMax technology is used in this study.)
 Abstract Checkpoint inhibitor (CPI) therapies provide limited benefit to patients with tumors of low immune reactivity. T cell-inducing vaccines hold promise to exert long-lasting disease control in combination with CPI therapy. Safety, tolerability and recommended phase 2 dose (RP2D) of an individualized, heterologous chimpanzee adenovirus (ChAd68) and self-amplifying mRNA (samRNA)-based neoantigen vaccine in combination with nivolumab and ipilimumab were assessed as primary endpoints in an ongoing phase 1/2 study in patients with advanced metastatic solid tumors (NCT03639714). The individualized vaccine regimen was safe and well tolerated, with no dose-limiting toxicities. Treatment-related adverse events (TRAEs) >10% included pyrexia, fatigue, musculoskeletal and injection site pain and diarrhea. Serious TRAEs included one count each of pyrexia, duodenitis, increased transaminases and hyperthyroidism. The RP2D was 1012 viral particles (VP) ChAd68 and 30 µg samRNA. Secondary endpoints included immunogenicity, feasibility of manufacturing and overall survival (OS). Vaccine manufacturing was feasible, with vaccination inducing long-lasting neoantigen-specific CD8 T cell responses. Several patients with microsatellite-stable colorectal cancer (MSS-CRC) had improved OS. Exploratory biomarker analyses showed decreased circulating tumor DNA (ctDNA) in patients with prolonged OS. Although small study size limits statistical and translational analyses, the increased OS observed in MSS-CRC warrants further exploration in larger randomized studies.
Abstract Checkpoint inhibitor (CPI) therapies provide limited benefit to patients with tumors of low immune reactivity. T cell-inducing vaccines hold promise to exert long-lasting disease control in combination with CPI therapy. Safety, tolerability and recommended phase 2 dose (RP2D) of an individualized, heterologous chimpanzee adenovirus (ChAd68) and self-amplifying mRNA (samRNA)-based neoantigen vaccine in combination with nivolumab and ipilimumab were assessed as primary endpoints in an ongoing phase 1/2 study in patients with advanced metastatic solid tumors (NCT03639714). The individualized vaccine regimen was safe and well tolerated, with no dose-limiting toxicities. Treatment-related adverse events (TRAEs) >10% included pyrexia, fatigue, musculoskeletal and injection site pain and diarrhea. Serious TRAEs included one count each of pyrexia, duodenitis, increased transaminases and hyperthyroidism. The RP2D was 1012 viral particles (VP) ChAd68 and 30 µg samRNA. Secondary endpoints included immunogenicity, feasibility of manufacturing and overall survival (OS). Vaccine manufacturing was feasible, with vaccination inducing long-lasting neoantigen-specific CD8 T cell responses. Several patients with microsatellite-stable colorectal cancer (MSS-CRC) had improved OS. Exploratory biomarker analyses showed decreased circulating tumor DNA (ctDNA) in patients with prolonged OS. Although small study size limits statistical and translational analyses, the increased OS observed in MSS-CRC warrants further exploration in larger randomized studies.
(Methods section) cfDNA was extracted from the entire plasma volume of a single draw using the Apostle MiniMax cfDNA Isolation kit (ApostleBio)
45. Simultaneous analysis of mutations and methylations in circulating cell-free DNA for hepatocellular carcinoma detection. Pei Wang, Qianqian Song, Jie Ren, et al. Science Translational Medicine 14, eabp8704 (2022) 23 November 2022
(Note: Apostle MiniMax technology is used in this study.)
 Cell-free DNA (cfDNA)–based liquid biopsy is a promising approach for the early detection of cancer. A major hurdle is the limited yield of cfDNA from one blood draw, limiting the use of most samples to one test of either mutation or methylation. Here, we develop a technology, Mutation Capsule Plus (MCP), which enables multiplex profiling of one cfDNA sample, including simultaneous detection of genetic and epigenetic alterations and genome-wide discovery of methylation markers. With this technology, we performed de novo screening of methylation markers on cfDNA samples from 30 hepatocellular carcinoma (HCC) cases and 30 non-HCC controls. The methylation markers enriched in HCC cfDNA were further profiled in parallel with a panel of mutations on a training cohort of 60 HCC and 60 non-HCC cases, resulting in an HCC detection model. We validated the model in an independent retrospective cohort with 58 HCC and 198 non-HCC cases and got 90% sensitivity with 94% specificity. Furthermore, we applied the model to a prospective cohort of 311 asymptomatic hepatitis B virus carriers with normal liver ultrasonography and serum AFP concentration. The model detected four of the five HCC cases in the cohort, showing 80% sensitivity and 94% specificity. These findings demonstrate that the MCP technology has potential for the discovery and validation of multiomics biomarkers for the noninvasive detection of cancer. This study also provides a comprehensive database of genetic and epigenetic alterations in the cfDNA of a large cohort of HCC cases and high-risk non-HCC individuals.
Cell-free DNA (cfDNA)–based liquid biopsy is a promising approach for the early detection of cancer. A major hurdle is the limited yield of cfDNA from one blood draw, limiting the use of most samples to one test of either mutation or methylation. Here, we develop a technology, Mutation Capsule Plus (MCP), which enables multiplex profiling of one cfDNA sample, including simultaneous detection of genetic and epigenetic alterations and genome-wide discovery of methylation markers. With this technology, we performed de novo screening of methylation markers on cfDNA samples from 30 hepatocellular carcinoma (HCC) cases and 30 non-HCC controls. The methylation markers enriched in HCC cfDNA were further profiled in parallel with a panel of mutations on a training cohort of 60 HCC and 60 non-HCC cases, resulting in an HCC detection model. We validated the model in an independent retrospective cohort with 58 HCC and 198 non-HCC cases and got 90% sensitivity with 94% specificity. Furthermore, we applied the model to a prospective cohort of 311 asymptomatic hepatitis B virus carriers with normal liver ultrasonography and serum AFP concentration. The model detected four of the five HCC cases in the cohort, showing 80% sensitivity and 94% specificity. These findings demonstrate that the MCP technology has potential for the discovery and validation of multiomics biomarkers for the noninvasive detection of cancer. This study also provides a comprehensive database of genetic and epigenetic alterations in the cfDNA of a large cohort of HCC cases and high-risk non-HCC individuals.
(Methods Section) cfDNA was extracted from the plasma samples using the Apostle MiniMax cfDNA isolation kit (C43468, Apostle).
44. Safety and tolerability of AAV8 delivery of a broadly neutralizing antibody in adults living with HIV: a phase 1, dose-escalation trial. Casazza JP, Cale EM, et al. the VRC603 Study Team. Nature Medicine April 11, 2022; https://www.nature.com/articles/s41591-022-01762-x
(Note: Apostle MiniMax technology is used in this study.)
 Adeno-associated viral vector-mediated transfer of DNA coding for broadly neutralizing anti-HIV antibodies (bnAbs) offers an alternative to attempting to induce protection by vaccination or by repeated infusions of bnAbs. In this study, we administered a recombinant bicistronic adeno-associated virus (AAV8) vector coding for both the light and heavy chains of the potent broadly neutralizing HIV-1 antibody VRC07 (AAV8-VRC07) to eight adults living with HIV. All participants remained on effective anti-retroviral therapy (viral load (VL) <50 copies per milliliter) throughout this phase 1, dose-escalation clinical trial (NCT03374202). AAV8-VRC07 was given at doses of 5 × 1010, 5 × 1011 and 2.5 × 1012 vector genomes per kilogram by intramuscular (IM) injection. Primary endpoints of this study were to assess the safety and tolerability of AAV8-VRC07; to determine the pharmacokinetics and immunogenicity of in vivo VRC07 production; and to describe the immune response directed against AAV8-VRC07 vector and its products. Secondary endpoints were to assess the clinical effects of AAV8-VRC07 on CD4 T cell count and VL and to assess the persistence of VRC07 produced in participants. In this cohort, IM injection of AAV8-VRC07 was safe and well tolerated. No clinically significant change in CD4 T cell count or VL occurred during the 1–3 years of follow-up reported here. In participants who received AAV8-VRC07, concentrations of VRC07 were increased 6 weeks (P = 0.008) and 52 weeks (P = 0.016) after IM injection of the product. All eight individuals produced measurable amounts of serum VRC07, with maximal VRC07 concentrations >1 µg ml−1 in three individuals. In four individuals, VRC07 serum concentrations remained stable near maximal concentration for up to 3 years of follow-up. In exploratory analyses, neutralizing activity of in vivo produced VRC07 was similar to that of in vitro produced VRC07. Three of eight participants showed a non-idiotypic anti-drug antibody (ADA) response directed against the Fab portion of VRC07. This ADA response appeared to decrease the production of serum VRC07 in two of these three participants. These data represent a proof of concept that adeno-associated viral vectors can durably produce biologically active, difficult-to-induce bnAbs in vivo, which could add valuable new tools to the fight against infectious diseases.
Adeno-associated viral vector-mediated transfer of DNA coding for broadly neutralizing anti-HIV antibodies (bnAbs) offers an alternative to attempting to induce protection by vaccination or by repeated infusions of bnAbs. In this study, we administered a recombinant bicistronic adeno-associated virus (AAV8) vector coding for both the light and heavy chains of the potent broadly neutralizing HIV-1 antibody VRC07 (AAV8-VRC07) to eight adults living with HIV. All participants remained on effective anti-retroviral therapy (viral load (VL) <50 copies per milliliter) throughout this phase 1, dose-escalation clinical trial (NCT03374202). AAV8-VRC07 was given at doses of 5 × 1010, 5 × 1011 and 2.5 × 1012 vector genomes per kilogram by intramuscular (IM) injection. Primary endpoints of this study were to assess the safety and tolerability of AAV8-VRC07; to determine the pharmacokinetics and immunogenicity of in vivo VRC07 production; and to describe the immune response directed against AAV8-VRC07 vector and its products. Secondary endpoints were to assess the clinical effects of AAV8-VRC07 on CD4 T cell count and VL and to assess the persistence of VRC07 produced in participants. In this cohort, IM injection of AAV8-VRC07 was safe and well tolerated. No clinically significant change in CD4 T cell count or VL occurred during the 1–3 years of follow-up reported here. In participants who received AAV8-VRC07, concentrations of VRC07 were increased 6 weeks (P = 0.008) and 52 weeks (P = 0.016) after IM injection of the product. All eight individuals produced measurable amounts of serum VRC07, with maximal VRC07 concentrations >1 µg ml−1 in three individuals. In four individuals, VRC07 serum concentrations remained stable near maximal concentration for up to 3 years of follow-up. In exploratory analyses, neutralizing activity of in vivo produced VRC07 was similar to that of in vitro produced VRC07. Three of eight participants showed a non-idiotypic anti-drug antibody (ADA) response directed against the Fab portion of VRC07. This ADA response appeared to decrease the production of serum VRC07 in two of these three participants. These data represent a proof of concept that adeno-associated viral vectors can durably produce biologically active, difficult-to-induce bnAbs in vivo, which could add valuable new tools to the fight against infectious diseases.
(Methods Section) AAV8-VRC07 vector DNA quantitation. Plasma AAV8-VRC07 plasmid DNA was measured by extracting DNA from plasma, concentrating and then using a real-time PCR assay to measure a 103 base sequence spanning the junction of the IgG heavy chain sequence and F2A insert. DNA was extracted from serum using an Apostle MiniMax High Efficiency cfDNA Isolation Kit, following the manufacturer’s protocol with slight modification.
43. Comparative analysis of cell-free DNA extraction efficiency from plasma. Beckman Coulter Life Sciences. Indianapolis, IN. Technical Note 2022.
(Note: Apostle MiniMax technology is compared and discussed in this study.)
Results and discussion
Mean cfDNA concentrations in the eluates were determined to be 8.30 ng/μL for samples extracted with the QIAamp kit and 11.15 ng/μL for samples extracted with the Apostle kit when measured with the Qubit assay (Figure 1A). This illustrates a 34.3% higher yield when using the Apostle kit as compared to the QIAamp kit.
When evaluating the contents of specific genes in the eluate, the Apostle purification displayed a higher yield than QIAamp (Figure 1B). 8358 DNAJC5G positive droplets were detected in the Apostle samples compared to 7269 droplets in the QIAamp samples. Similarly, 10849 ACTG1 droplets were detected in the Apostle-extracted samples versus 9463 droplets detected in the QIAamp-extracted samples. This data demonstrates an average of 14.8% higher yield using the Apostle kit.
Finally, when evaluating cfDNA fragments via the Agilent bioanalyzer instrument, the Apostle kit demonstrated the highest yield (Figure 1C and D). Mononucleosomal cfDNA concentration in the eluate of Apostle samples was estimated to be 10.64 ng/μL versus 6.26 ng/μL in the eluate of QIAamp samples. The dinucleosomal cfDNA concentration was low for both kits, with 0.13 ng/μL recorded for Apostle samples and 0.24 ng/μL for QIAamp samples. This indicates a slightly better purification of dinucleosomal cfDNA using QIAamp; however, this could also be an artifact given the low concentrations. The Apostle mononucleosomal cfDNA yield was 70.0% higher than the yield for the QIAamp samples.
A summary provided in Table 2 shows the consistently higher yield achieved by the Apostle MiniMax High Efficiency cfDNA Isolation Kit as compared to the QIAamp® circulating nucleic acid kit. The average 40% improvement in yield underscores the importance of extraction kit selection in cfDNA assay development and highlights the potential impact of cfDNA isolation efficiency in ctDNA detection.
42. Reliability of Cell-Free DNA and Targeted NGS in Predicting Chromosomal Abnormalities of Patients With Myeloid Neoplasms. Andrew Ip, Alexandra Della Pia, Gee Youn (Geeny) Kim, Jason Lofters, James Behrmann, Dylon Patel, Simone Kats, Jeffrey Justin Estella, Ivan De Dios, Wanlong Ma, Andrew L. Pecora, Andre H. Goy, Jamie Koprivnikar, James K. McCloskey and Maher Albitar. Frontiers in Oncology June 14, 2022; https://doi.org/10.3389/fonc.2022.923809
(Note: Apostle MiniMax technology is used in this study.)
 Introduction: Cytogenetic analysis is important for stratifying patients with various neoplasms. We explored the use of targeted next generation sequencing (NGS) in detecting chromosomal structural abnormalities or copy number variations (CNVs) in patients with myeloid neoplasms.
Introduction: Cytogenetic analysis is important for stratifying patients with various neoplasms. We explored the use of targeted next generation sequencing (NGS) in detecting chromosomal structural abnormalities or copy number variations (CNVs) in patients with myeloid neoplasms.
Methods: Plasma cell-free DNA (cfDNA) from 2821 myeloid or lymphoid neoplasm patients were collected. cfDNA was sequenced using a 275 gene panel. CNVkit software was used for analyzing and visualizing CNVs. Cytogenetic data from corresponding bone marrow (BM) samples was available on 89 myeloid samples.
Results: Of the 2821 samples, 1539 (54.5%) showed evidence of mutations consistent with the presence of neoplastic clones in circulation. Of these 1539 samples, 906 (59%) showed abnormalities associated with myeloid neoplasms and 633 (41%) with lymphoid neoplasms. Chromosomal structural abnormalities in cfDNA were detected in 146 (16%) myeloid samples and 76 (12%) lymphoid samples. Upon comparison of the myeloid samples with 89 BM patients, NGS testing was able to reliably detect chromosomal gain or loss, except for fusion abnormalities. When cytogenetic abnormalities were classified according to prognostic classes, there was a complete (100%) concordance between cfDNA NGS data and cytogenetic data.
Conclusions: This data shows that liquid biopsy using targeted NGS is reliable in detecting chromosomal structural abnormalities in myeloid neoplasms. In specific circumstances, targeted NGS may be reliable and efficient to provide adequate information without the need for BM biopsy considering broad mutation profiling can be obtained through adequate sequencing within the same test. Overall, this study supports the use of liquid biopsy for early diagnosis and monitoring of patients with myeloid neoplasms.
(Methods Section) Sample Collection and cfDNA Isolation
We extracted cfDNA from 2821 peripheral blood samples using the Apostle MiniMax(TM) High Efficiency cfDNA Isolation Kit (San Jose, CA). These peripheral blood samples were collected in EDTA anticoagulant over approximately 18 months. DNA was extracted from separated plasma within 48 hours of collection. We tested these samples for the presence of circulating tumor DNA (ctDNA).
41. Identification of multi-omic biomarkers from Fecal DNA for improved Detection of Colorectal Cancer and precancerous lesions. Yujing Fang, Jiaxi Peng, Zhilong Li, et al. medRxiv. November 13, 2022. https://doi.org/10.1101/2022.11.08.22282099
(Note: Apostle MiniGenomics technology is used in this study.)

Background Timely diagnosis and intervention of colorectal cancer (CRC) at curable stages is essential for improving patient survival. Stool samples carry exfoliation of intestinal epithelium, therefore providing excellent opportunity for non-invasive diagnosis of CRC as well as precancerous lesions. In this study, we aimed to conduct multi-dimensional analysis of fecal DNA and investigate the utility of different types of biomarkers for CRC detection.
Method In this case-control study, we performed comprehensive analyses of the genomic, epigenomic, and metagenomic features of fecal DNA from CRC patients, individuals with advanced precancerous lesions (APLs) and controls. DNA methylation markers were identified by whole genome bisulfite sequencing of paired colorectal cancer and normal tissues. A multi-gene fecal DNA methylation test was then developed based on three marker genes (SDC2, ADHFE1 and PPP2R5C) using quantitative methylation-specific PCR (qMSP), and validated on fecal DNA samples. Genomic mutation profiles as well as microbiome signatures of fecal DNA were analyzed using high-throughput sequencing.
Results The methylation-based fecal DNA test demonstrated an overall sensitivity of 88% for CRC and 46.2% for APL respectively, and a specificity of 91.8% for controls. On the other hand, the mutation-based diagnostic model yielded limited sensitivity, and combined detection of methylation markers and mutation in fecal DNA did not improve the assay performance. Meanwhile, a diagnostic model based on the relative abundance of bacterial species showed inferior performance than the methylation-based model. Finally, integrated diagnostic model combining both methylation and microbial markers showed an enhanced performance (AUC= 0.95) compared to methylation markers alone.
Conclusions The multi-gene fecal DNA methylation test provided remarkable diagnostic performance for CRCs and APLs. Furthermore, multi-target assay integrating both methylation and microbial markers may further improve the diagnostic performance. Our findings may aid in the development of novel diagnostic tools for CRC.
(Methods section)
For stool samples, fecal DNA was isolated by Apostle MiniGenomics Stool Fast Kit (Apostle, A181206).
40. Quantifying Fetal DNA in Maternal Blood Plasma by ddPCR Using DNA Methylation. E. Hall, T. Riel, M. Ramesh, M. Gencoglu, O. Mikhaylichenko, R. Dannebaum, S. Margeridon, M. Herrera. Bio-Rad Laboratories, Pleasanton, CA. Association for Molecular Pathology 2022 Annual Meeting Abstracts. J Mol Diagn 2022, 24:S1 Abstract G072. (Full Abstract List) (G072 Only)
(Note: Apostle MiniMax technology is used in this study.)
 Introduction: The proportion of cell-free DNA (cfDNA) circulating in maternal blood that originates from the fetus, the fetal fraction, is an important quality control metric when performing tests on fetal-derived cfDNA. Epigenetic differences produce dissimilar DNA methylation patterns, allowing for leveraging regions of high methylation contrast using methylation-sensitive restriction enzyme (MSRE) digestion to quantify fetal and maternal DNA via droplet digital PCR (ddPCR). This advancement positions ddPCR as a faster and less expensive alternative to next-generation sequencing (NGS) for fetal fraction estimation.
Introduction: The proportion of cell-free DNA (cfDNA) circulating in maternal blood that originates from the fetus, the fetal fraction, is an important quality control metric when performing tests on fetal-derived cfDNA. Epigenetic differences produce dissimilar DNA methylation patterns, allowing for leveraging regions of high methylation contrast using methylation-sensitive restriction enzyme (MSRE) digestion to quantify fetal and maternal DNA via droplet digital PCR (ddPCR). This advancement positions ddPCR as a faster and less expensive alternative to next-generation sequencing (NGS) for fetal fraction estimation.
Methods: Assays were designed to target MSRE- compatible regions with high methylation contrast between maternal and fetal cfDNA. Fetal assays targeted sites hypermethylated in fetal cfDNA and maternal assays targeted sites hypermethylated in maternal cfDNA. The assay multiplex was tested against contrived and clinical samples using an in-droplet MSRE-ddPCR workflow. The reaction mix was dropletized to create about 20,000 droplets per 24-μL reaction, thermocycled, and analyzed in a QX ONE instrument. The thermocycling profile included a 45-minute MSRE incubation step prior to PCR amplification. Contrived samples were constructed by spiking DNA-free plasma with micrococcal nuclease-digested DNA from an amniotic fluid cell line ("fetal" component) and a B-lymphocyte cell line ("maternal" component). Clinical samples were remnant diagnostic samples with existing NGS non-invasive prenatal testing results attached. DNA was extracted from all samples with the Apostle MiniMax kit on the KingFisher Flex.
Results: From an initial set of 15 assays, a final five-assay multiplex was produced following amplicon sequencing with NGS and ddPCR screening. Although amplicon sequencing did not completely predict ddPCR performance and non- specific interactions, it was valuable for guiding the final ddPCR screen. The five-assay multiplex, consisting of three fetal assays and two maternal assays, produced an excellent linear response against contrived samples from 0% to 25% fetal fraction (R2 >0.99). Similarly, a high correlation was observed between ddPCR-estimated fetal fraction and NGS fetal fraction for a set of clinical samples (n=6, plus two non- pregnant controls, R2 >0.94).
Conclusions: As an epigenetic trait, DNA methylation is a useful way to discriminate between otherwise highly similar DNA sequences in an efficient and effective manner. Leveraging DNA methylation may be done with minimal impact to the standard ddPCR workflow. The high sensitivity, speed, and direct quantification of ddPCR make it an attractive alternative to NGS for fetal fraction estimation.
39. Next-Generation Digital Polymerase Chain Reaction: High-Dynamic-Range Single-Molecule DNA Counting via Ultrapartitioning. Eleen Y. Shum, Janice H. Lai, Sixing Li, Haeun G. Lee, Jesse Soliman, Vedant K. Raol, Cavina K. Lee, Stephen P.A. Fodor, H. Christina Fan Anal. Chem. 2022, Publication Date:December 12, 2022 https://doi.org/10.1021/acs.analchem.2c03649
(Note: Apostle MiniMax technology is used in this study.)
 ABSTRACT: Digital PCR (dPCR) was first conceived for single-molecule quantitation. However, current dPCR systems often require DNA templates to share partitions due to limited partitioning capacities. Here, we introduce UltraPCR, a next-generation dPCR system where DNA counting is performed in a single-molecule regimen through a 6-log dynamic range using a swift and parallelized workflow. Each UltraPCR reaction is divided into >30 million partitions without microfluidics to achieve single template occupancy. Combined with a unique emulsion chemistry, partitions are optically clear, enabling the use of a three-dimensional imaging technique to rapidly detect DNA-positive partitions. Single-molecule occupancy also allows for more straightforward multiplex assay development due to the absence of partition-specific competition. As a proof of concept, we developed a 222-plex UltraPCR assay and demonstrated its potential use as a rapid, low-cost screening assay for noninvasive prenatal testing for as low as 4% trisomy fraction samples with high precision, accuracy, and reproducibility.
ABSTRACT: Digital PCR (dPCR) was first conceived for single-molecule quantitation. However, current dPCR systems often require DNA templates to share partitions due to limited partitioning capacities. Here, we introduce UltraPCR, a next-generation dPCR system where DNA counting is performed in a single-molecule regimen through a 6-log dynamic range using a swift and parallelized workflow. Each UltraPCR reaction is divided into >30 million partitions without microfluidics to achieve single template occupancy. Combined with a unique emulsion chemistry, partitions are optically clear, enabling the use of a three-dimensional imaging technique to rapidly detect DNA-positive partitions. Single-molecule occupancy also allows for more straightforward multiplex assay development due to the absence of partition-specific competition. As a proof of concept, we developed a 222-plex UltraPCR assay and demonstrated its potential use as a rapid, low-cost screening assay for noninvasive prenatal testing for as low as 4% trisomy fraction samples with high precision, accuracy, and reproducibility.
(Methods Section) Cell-free plasma was isolated using the Apostle MiniMax kit (Beckman Coulter) according to the manufacturer’s protocol with an elution volume of 60 μL.
38. A method for early diagnosis of lung cancer from tumor originated DNA fragments using plasma cfDNA methylome and fragmentome profiles. Yeo Jin Kim, Hahyeon Jeona, Sungwon Jeon, et al. Molecular and Cellular Probes Volume 66, December 2022
(Note: Apostle MiniMax technology is used in this study.)
 Abstract
Abstract
Early detection is critical for minimizing mortality from cancer. Plasma cell-free DNA (cfDNA) contains the signatures of tumor DNA, allowing us to quantify the signature and diagnose early-stage tumors. Here, we report a novel tumor fragment quantification method, TOF (Tumor Originated Fragment) for the diagnosis of lung cancer by quantifying and analyzing both the plasma cfDNA methylation patterns and fragmentomic signatures. TOF utilizes the amount of ctDNA predicted from the methylation density information of each cfDNA read mapped on 6243 lung-tumor-specific CpG markers. The 6243 tumor-specific markers were derived from lung tumor tissues by comparing them with corresponding normal tissues and healthy blood from public methylation data. TOF also utilizes two cfDNA fragmentomic signatures: 1) the short fragment ratio, and 2) the 5′ end-motif profile. We used 298 plasma samples to analyze cfDNA signatures using enzymatic methyl-sequencing data from 201 lung cancer patients and 97 healthy controls. The TOF score showed 0.98 of the area under the curve in correctly classifying lung cancer from normal samples. The TOF score resolution was high enough to clearly differentiate even the early-stage non-small cell lung cancer patients from the healthy controls. The same was true for small cell lung cancer patients.
(Methods Section) Cell-free DNA was extracted from 3 to 5 ml of plasma using XXX method or Apostle MiniMaxTM High Efficiency Isolation Kit (Beckman Coulter Life Sciences, C40603), according to the manufacturers’ procedures.
37. Nicotine dose-dependent epigenomic-wide DNA methylation changes in the mice with long-term electronic cigarette exposure. Gang Peng,* Yibo Xi,* Chiara Bellini, Kien Pham, Zhen W Zhuang, Qin Yan, Man Jia, Guilin Wang, Lingeng Lu, Moon-Shong Tang, Hongyu Zhao, and He Wang American Journal of Cancer Research. Volume 12, Issue 8, August 2022, Pages 3679–3692.
(Note: Apostle MiniMax technology is us in this study.)
 Abstract
Abstract
Epigenomic-wide DNA methylation profiling holds the potential to reflect both electronic cigarette exposure-associated risks and individual poor health outcomes. However, a systemic study in animals or humans is still lacking. Using the Infinium Mouse Methylation BeadChip, we examined the DNA methylation status of white blood cells in male ApoE-/- mice after 14 weeks of electronic cigarette exposure with the InExpose system (2 hr/day, 5 days/week, 50% PG and 50% VG) with low (6 mg/ml) and high (36 mg/ml) nicotine concentrations. Our results indicate that electronic cigarette aerosol inhalation induces significant alteration of 8,985 CpGs in a dose-dependent manner (FDR<0.05); 7,389 (82.2%) of the CpG sites are annotated with known genes. Among the top 6 significant CpG sites (P-value<1e-8), 4 CpG sites are located in the known genes, and most (3/5) of these genes have been related to cigarette smoking. The other two CpGs are close to/associated with the Phc2 gene that was recently linked to smoking in a transcriptome-wide associations study. Furthermore, the gene set enrichment analysis highlights the activation of MAPK and 4 cardiomyocyte/cardiomyopathy-related signaling pathways (including adrenergic signaling in cardiomyocytes and arrhythmogenic right ventricular cardiomyopathy) following repeated electronic cigarette use. The MAPK pathway activation correlates well with our finding of increased cytokine mRNA expression after electronic cigarette exposure in the same batch of mice. Interestingly, two pathways related to mitochondrial activities, namely mitochondrial gene expression and mitochondrial translation, are also activated after electronic cigarette exposure. Elucidating the relationship between these pathways and the increased circulating mitochondrial DNA observed here will provide further insight into the cell-damaging effects of prolonged inhalation of e-cigarette aerosols.
(Methods section) Plasma cfDNA and mtDNA/nDNA ratio and oxidization of cell-free DNA
For measurement of in vivo plasma mtDNA/nDNA ratio, mice plasma was first subjected to cell-free DNA (cfDNA) extraction using Apostle minimax cfDNA extraction kit (ApostleBio, CA, US).
36. High-throughput sample processing for methylation analysis in an automated, enclosed environment. Alejandro Stark, Thomas R. Pisanic, James G. Herman, Tza-Huei Wang. SLAS Technology Volume 27, Issue 3, June 2022, Pages 172-179.
(Note: Apostle MiniMax technology is compared and discussed in this study.)
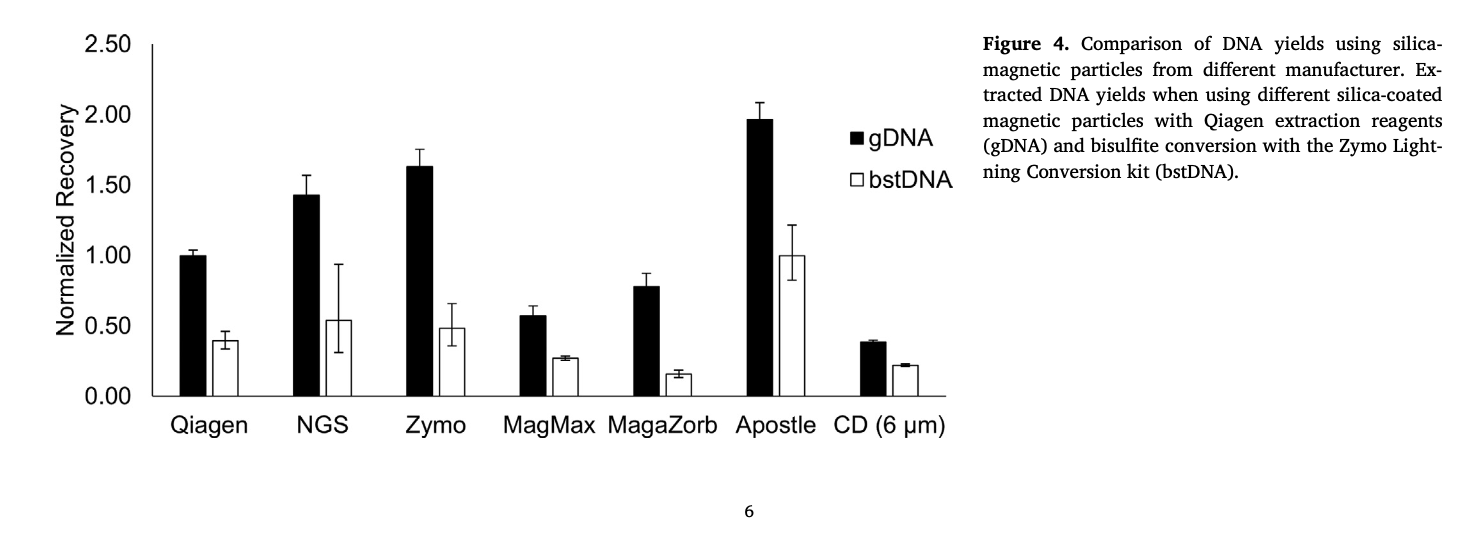
(Figure 4) - "Most notably, the Apostle particles outperformed all others, achieving almost 2-fold higher recovery yields than the particles supplied in the X kit. "
Variation in methylcytosine is perhaps the most well-studied epigenetic mechanism of gene regulation. Methods that have been developed and implemented for assessing DNA methylation require sample DNA to be extracted, purified and chemically-processed through bisulfite conversion before downstream analysis. While some automated solutions exist for each of these individual process steps, a fully integrated solution for accomplishing the entire process in a high-throughput manner has yet to be demonstrated. Thus, sample processing methods still require numerous manual steps that may reduce sample throughput and precision, while increasing the risk of contamination and human error. In this work, we present an integrated, automated solution for performing the entire sample preparation process, including DNA extraction, purification, bisulfite conversion and PCR plate preparation within in an enclosed environment. The method employs silica-coated magnetic particles that eliminate the need for a centrifuge or vacuum manifold, thereby reducing the complexity and cost of the required automation platform. Toward this end, we also compare commercial DNA extraction and bisulfite conversion kits to identify a protocol suitable for automation to significantly improve genomic and bisulfite-treated DNA yields over manufacturer protocols. Overall, this research demonstrated development of an automated protocol that offers the ability to generate high-quality, bisulfite-treated DNA samples in a high-throughput and clean environment with minimal user intervention and comparable yields to manual processing.
35. Detecting SARS-CoV-2 variants in wastewater and their correlation with circulating variants in the communities. Lin Li, Timsy Uppal, Paul D. Hartley, Andrew Gorzalski, Mark Pandori, Michael A. Picker, Subhash C. Verma & Krishna Pagilla. Scientific Reports volume 12, Article number: 16141 (2022) , September 2022
(Note: Apostle MiniGenomics technology is used in this study.)
Abstract
Detection of SARS-CoV-2 viral load in wastewater has been highly informative in estimating the approximate number of infected individuals in the surrounding communities. Recent developments in wastewater monitoring to determine community prevalence of COVID-19 further extends into identifying SARS-CoV-2 variants, including those being monitored for having enhanced transmissibility. We sequenced genomic RNA derived from wastewater to determine the variants of coronaviruses circulating in the communities. Wastewater samples were collected from Truckee Meadows Water Reclamation Facility (TMWRF) from November 2020 to June 2021. SARS-CoV-2 variants resulting from wastewater were compared with the variants detected in infected individuals' clinical specimens (nasal/nasopharyngeal swabs) during the same period and found conclusively in agreement. Therefore, wastewater monitoring for SARS-CoV-2 variants in the community is a feasible strategy as a complementary tool to clinical specimen testing in the latter's absence.
(Methods Section) The NP swabs received at the Nevada State Public Health Laboratory (NSPHL) from the Reno-Sparks metropolitan for SARS-CoV-2 diagnostic testing were subjected for RNA extraction using Virus Total NA Isolation Fast Kit (Apostle MiniGenomics, San Jose, CA, USA), followed by RT-PCR for the detection of SARS-CoV-2 genome using US Federal Drug Administration Emergency Use Authorization (FDA-EUA) diagnostic kits, as described previously.
34. Optimalizace digitální polymerázové řetězové reakce pro aplikaci v neinvazivní prenatální diagnostice (Optimization of digital polymerase chain reaction for application in non-invasive prenatal diagnostics) Author: Šenkyřík, Pavel; Advisor: Korabečná, Marie; Referee: Vodička, Radek; Faculty / Institute: Faculty of Science; Discipline: Anthropology and Human Genetics; Department: Department of Anthropology and Human Genetics; Date of defense: 13. 9. 2022; Publisher: Univerzita Karlova, Přírodovědecká fakulta; Language: Czech
(Note: Apostle MiniMax technology is discussed in this diploma thesis.)
(Note: Table 13 referenced below in this diploma thesis shows that Apostle MiniMax yields the greatest number of analyzable drops in the digital PCR.)
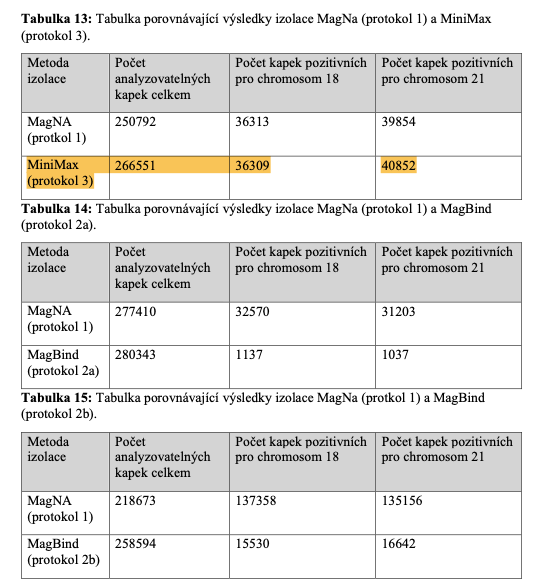
33. Rapid repeat infection of SARS-CoV-2 by two highly distinct delta-lineage viruses. Andrew J. Gorzalski , Christina Boyles, Victoria Sepcic, Subhash Verma, Joel Sevinsky, Kevin Libuit, Stephanie Van Hooser, Mark W. Pandori. Diagnostic Microbiology and Infectious Disease Volume 104, Issue 1, September 2022, 115747; https://doi.org/10.1016/j.diagmicrobio.2022.115747
(Note: Apostle MagTouch technology is used in this study.)
 An instance of sequential infection of an individual with, firstly, the Delta variant and secondly a Delta-sub-lineage has been identified. The individual was found positive for the AY.26 lineage 22 days after being found positive for the Delta [B.1.617.2] variant. The viruses associated with the cases showed dramatic genomic difference, including 31 changes that resulted in deletions or amino acid substitutions. Seven of these differences were observed in the Spike protein. The patient in question was between 30 and 35 years old and had no underlying health conditions. Though singular, this case illustrates the possibility that infection with the Delta variant may not itself be fully protective against a population of SARS-CoV-2 variants that are becoming increasingly diverse.
An instance of sequential infection of an individual with, firstly, the Delta variant and secondly a Delta-sub-lineage has been identified. The individual was found positive for the AY.26 lineage 22 days after being found positive for the Delta [B.1.617.2] variant. The viruses associated with the cases showed dramatic genomic difference, including 31 changes that resulted in deletions or amino acid substitutions. Seven of these differences were observed in the Spike protein. The patient in question was between 30 and 35 years old and had no underlying health conditions. Though singular, this case illustrates the possibility that infection with the Delta variant may not itself be fully protective against a population of SARS-CoV-2 variants that are becoming increasingly diverse.
32. Response prediction and risk stratification of patients with rectal cancer after neoadjuvant therapy through an analysis of circulating tumour DNA. Liu W, Li Y, et al. EBioMedicine March 17, 2022; https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(22)00129-3/fulltext
(Note: Apostle MiniMax technology is used in this study.)
 Background - Multiple approaches based on cell-free DNA (cfDNA) have been applied to detect minimal residual disease (MRD) and to predict prognosis or recurrence. However, a comparison of the approaches used in different cohorts and studies is difficult. We aimed to compare multiple approaches for MRD analysis after neoadjuvant therapy (NAT) in patients with locally advanced rectal cancer (LARC).
Background - Multiple approaches based on cell-free DNA (cfDNA) have been applied to detect minimal residual disease (MRD) and to predict prognosis or recurrence. However, a comparison of the approaches used in different cohorts and studies is difficult. We aimed to compare multiple approaches for MRD analysis after neoadjuvant therapy (NAT) in patients with locally advanced rectal cancer (LARC).
Methods - Sixty patients with LARC from a multicentre, phase II/III randomized trial were included, with tissue and blood samples collected. For each cfDNA sample, we profiled MRD using 3 approaches: personalized assay targeting tumour-informed mutations, universal panel of genes frequently mutated in colorectal cancer (CRC), and low depth sequencing for copy number alterations (CNAs).
Findings - Positive MRD based on post-NAT personalized assay was significantly associated with an increased risk of recurrence (HR = 27.38; log-rank P < 0.0001). MRD analysis based on universal panel (HR = 5.18; log-rank P = 0.00086) and CNAs analysis (HR = 9.24; log-rank P = 0.00017) showed a compromised performance in predicting recurrence. Both the personalized assay and universal panel showed complementary pattern to CNAs analysis in detecting cases with recurrence and the combination of the two types of biomarkers may lead to better performance.
Interpretation - The combination of mutation profiling and CNA profiling can improve the detection of MRD, which may help optimize the treatment strategies for patients with LARC.
31. A microfluidic platform for high-purity cell free DNA extraction from plasma for non-invasive prenatal testing. Lindsay Schneider, Thomas Usherwood, Anubhav Tripathi. Prenatal Diagnosis January 14, 2022; https://doi.org/10.1002/pd.6092
(Note: Apostle MiniMax technology is discussed in this study.)
 Objectives
Objectives
Increase the yield and purity of cell-free DNA (cfDNA) extracted from plasma for non-invasive prenatal testing (NIPT) as inefficiencies in this extraction and purification can dramatically affect the sensitivity and specificity of the test.
Methods
This work integrates cfDNA extraction from plasma with a microfluidic chip platform by combining magnetic bead-based extraction and electroosmotic flow on the microfluidic chip. Various wash buffers and voltage conditions were simulated using COMSOL Multiphysics Modeling and tested experimentally.
Results
When performing the first wash step of this assay on the microfluidic chip with 300 V applied across the channel there was a six-fold increase in the A260/A230 ratio showing a significant improvement (p value 0.0005) in the purity of the extracted sample all while maintaining a yield of 68.19%. These values are critical as a high yield results in more sample to analyze and an increase in A260/A230 ratio corresponds to a decrease in salt contaminants such as guanidinium thiocyanate which can interfere with downstream processes during DNA library preparation and potentially hinder the NIPT screening results.
Conclusions
This technique has the potential to improve NIPT outcomes and other clinically relevant workflows that use cfDNA as an analyte such as cancer detection.
30. Direct capture and sequencing reveal ultra-short single-stranded DNA in biofluids. Cheng et al. iScience. 2022; In Press.
(Note: Apostle MiniMax technology is discussed in this study.)
 Summary
Summary
Cell-free DNA (cfDNA) has become the predominant analyte of liquid biopsy; however, recent studies suggest the presence of subnucleosomal-sized DNA fragments in circulation that are likely single-stranded. Here, we report a method called Direct Capture and Sequencing (DCS) tailored to recover such fragments from biofluids by directly capturing them using short degenerate probes followed by single strand-based library preparation and next generation sequencing. DCS revealed a new DNA population in biofluids, named ultra-short single-stranded DNA (ussDNA). Evaluation of the size distribution and abundance of ussDNA manifested generality of its presence in human, animal species, and plants. In human, red blood cells were found to contain abundant ussDNA; plasma-derived ussDNA exhibited modal size at 50nt. This work reports the presence of an understudied DNA population in circulation, and yet more work is awaiting to study its generation mechanism, tissue of origin, disease implications, etc.
29. Nanostructures in non-invasive prenatal genetic screening. Samira Sadeghi, Mahdi Rahaie & Bita Ostad-Hasanzadeh. Biomedical Engineering Letters volume 12, pages 3–18 (2022)
(Note: Apostle MiniEnrich technology is reviewed in this review article.)
 Abstract
Abstract
Prenatal screening is an important issue during pregnancy to ensure fetal and maternal health, as well as preventing the birth of a defective fetus and further problems such as extra costs for the family and society. The methods for the screening have progressed to non-invasive approaches over the recent years. Limitations of common standard screening tests, including invasive sampling, high risk of abortion and a big delay in result preparation have led to the introduction of new rapid and non-invasive approaches for screening. Non-invasive prenatal screening includes a wide range of procedures, including fetal cell-free DNA analysis, proteome, RNAs and other fetal biomarkers in maternal serum. These biomarkers require less invasive sampling than usual methods such as chorionic villus sampling, amniocentesis or cordocentesis. Advanced strategies including the development of nanobiosensors and the use of special nanoparticles have provided optimization and development of NIPS tests, which leads to more accurate, specific and sensitive screening tests, rapid and more reliable results and low cost, as well. This review discusses the specifications and limitations of current non-invasive prenatal screening tests and introduces a novel collection of detection methods reported studies on nanoparticles’ aided detection. It can open a new prospect for further studies and effective investigations in prenatal screening field.
(pp. 13) In another study, Zhang, et al. designed a magnetic nano-platform named MiniEnrich, used to enrich the cff-DNA fragments in maternal plasma with high resolution by developing two different magnetic nanoparticle types. It was suggested that the mentioned platform had a fair potential to help detect monogenic disorders in non-invasive prenatal screening [108].
28. Monitoring and Identifying Insulin-Like Growth Factor 1 Variants by Liquid Chromatography–High-Resolution Mass Spectrometry in a Clinical Laboratory. Ievgen Motorykin, Allison Li & Zengru Wu. Clinical Applications of Mass Spectrometry in Biomolecular Analysis pp 239–251. (Book chapter) 2022. Part of the Methods in Molecular Biology book series (MIMB,volume 2546)
(Note: Apostle MiniMax technology is used in this study.)
Publications (2021)
27. Segmental duplication as potential biomarkers for non-invasive prenatal testing of aneuploidies. Xinwen Chen, Yifan Li, Qiuying Huang, Xingming Lin, Xudong Wang, Yafang Wang, Ying Liu, Qiushun He, Yinghua Liu, Ting Wang, Zhi-Liang Ji, Qingge Li. EBioMedicine August 11, 2021; https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(21)00328-5/fulltext
(Note: Apostle MiniMax technology is used in this study.)
 We developed a computational program whereby available SD regions can be processed and analyzed efficiently for their potential use as biomarkers of the aneuploidy of interest. For the five common aneuploidies, i.e., trisomy 13, 18, 21, and two sex chromosome aneuploidies, a total of 21,772 candidate SD biomarker sequences together with their corresponding primer/probe sets were generated. The primer/probe sets were tested using a real-time PCR-based multicolour melting curve analysis for simultaneous detection of the five common aneuploidies, and yielded 100% clinical sensitivity and 99.64% specificity when subjected to a clinical evaluation. Following the observations that the SD biomarkers for aneuploidy could be better detected by digital PCR with improved accuracy, we established a noninvasive prenatal testing protocol for trisomy 21 and attained 100% concordance with next generation sequencing.
We developed a computational program whereby available SD regions can be processed and analyzed efficiently for their potential use as biomarkers of the aneuploidy of interest. For the five common aneuploidies, i.e., trisomy 13, 18, 21, and two sex chromosome aneuploidies, a total of 21,772 candidate SD biomarker sequences together with their corresponding primer/probe sets were generated. The primer/probe sets were tested using a real-time PCR-based multicolour melting curve analysis for simultaneous detection of the five common aneuploidies, and yielded 100% clinical sensitivity and 99.64% specificity when subjected to a clinical evaluation. Following the observations that the SD biomarkers for aneuploidy could be better detected by digital PCR with improved accuracy, we established a noninvasive prenatal testing protocol for trisomy 21 and attained 100% concordance with next generation sequencing.
Our study confirmed that SD regions are preferred biomarkers for aneuploidy detection and in particular SD-based digital PCR could find potential use for NIPT of trisomy. A similar strategy can be applied to other chromosomal abnormality and genetic disorders.
26. Isotopic Peak Index, Relative Retention Time, and Tandem MS for Automated High Throughput IGF-1 Variants Identification in a Clinical Laboratory. Ievgen Motorykin, Hua Li, Nigel J. Clarke, Michael J. McPhaul, and Zengru Wu. Analytical Chemistry August 2021. https://doi.org/10.1021/acs.analchem.1c02566
(Note: Apostle MiniMax technology is used in this study.)
Measuring insulin-like growth factor-1 (IGF-1) is useful for assessing and managing growth-related disorders, such as acromegaly and growth hormone deficiency. High-resolution liquid chromatography–mass spectrometry (LC–MS) is used for measuring IGF-1 due to its molecular specificity, quantitative performance, well-characterized reference materials, and detailed age/sex-specific reference intervals. However, polymorphisms in the IGF1 gene may cause mass shifts in the polypeptide, which can impede quantitation and cause errors in clinical interpretation. We (1) developed a concept of “isotopic peak index”, which allows simultaneous monitoring of 15 IGF-1 variants by using only four m/z ratios; (2) developed a “relative retention time” parameter that allows distinction of previously unresolved variants; and (3) utilized tandem mass spectrometry (MS/MS) to distinguish between the most common pair of variants: isobaric A67T and A70T. All methods were validated with DNA sequencing. This approach identified six variants from the ExAC database, P66A, A67S, S34N, A38 V, A67T, and A70T; two previously reported V44M and A67V variants; and discovered six unreported variants, Y31H, S33P, R50Q, R56K, T41I, and A62T. Major improvements in our workflow include enhanced automation, avoiding detailed manual calculations that are prone to human error, and the ability to monitor more, and discover new, IGF-1 variants. The workflow provides a profile of a patient’s IGF-1 status and can be used to explore genotype–phenotype relationships in IGF-1 variants.
25. FDA EUA Summary: Fulgent COVID-19 by RT-PCR TEST(FULGENT THERAPEUTICS). For In vitro Diagnostic Use. Rx Only. For use under Emergency Use Authorization (EUA) only. US FDA. April 12, 2021.
(Note: Apostle RNA Extraction Method is included in this US FDA EUA.)
Weblink for the US FDA EUA letter: https://www.fda.gov/media/138147/download
24. Efficient detection and post-surgical monitoring of colon cancer with a multi-marker DNA methylation liquid biopsy. Shengnan Jin, Dewen Zhu, Fanggui Shao, Shiliang Chen, Ying Guo, Kuan Li, Yourong Wang, Rongxiu Ding, Lingjia Gao, Wen Ma, Tong Lu, Dandan Li, Zhengzheng Zhang, Suili Cai, Xue Liang, Huayu Song, Ling Ji, Jinlei Li, Zhihai Zheng, Feizhao Jiang, Xiaoli Wu, Ju Luan, Huxiang Zhang, Zhengquan Yang, Charles R. Cantor, Chang Xu, and Chunming Ding. PNAS February 2, 2021 118 (5) e2017421118; https://doi.org/10.1073/pnas.2017421118
(Note: Apostle MiniMax technology is used in this study.)
 Multiplex assays, involving the simultaneous use of multiple circulating tumor DNA (ctDNA) markers, can improve the performance of liquid biopsies so that they are highly predictive of cancer recurrence. We have developed a single-tube methylation-specific quantitative PCR assay (mqMSP) that uses 10 different methylation markers and is capable of quantitative analysis of plasma samples with as little as 0.05% tumor DNA. In a cohort of 179 plasma samples from colorectal cancer (CRC) patients, adenoma patients, and healthy controls, the sensitivity and specificity of the mqMSP assay were 84.9% and 83.3%, respectively. In a head-to-head comparative study, the mqMSP assay also performed better for detecting early-stage (stage I and II) and premalignant polyps than a published SEPT9 assay. In an independent longitudinal cohort of 182 plasma samples (preoperative, postoperative, and follow-up) from 82 CRC patients, the mqMSP assay detected ctDNA in 73 (89.0%) of the preoperative plasma samples. Postoperative detection of ctDNA (within 2 wk of surgery) identified 11 of the 20 recurrence patients and was associated with poorer recurrence-free survival (hazard ratio, 4.20; P = 0.0005). With subsequent longitudinal monitoring, 14 patients (70%) had detectable ctDNA before recurrence, with a median lead time of 8.0 mo earlier than seen with radiologic imaging. The mqMSP assay is cost-effective and easily implementable for routine clinical monitoring of CRC recurrence, which can lead to better patient management after surgery.
Multiplex assays, involving the simultaneous use of multiple circulating tumor DNA (ctDNA) markers, can improve the performance of liquid biopsies so that they are highly predictive of cancer recurrence. We have developed a single-tube methylation-specific quantitative PCR assay (mqMSP) that uses 10 different methylation markers and is capable of quantitative analysis of plasma samples with as little as 0.05% tumor DNA. In a cohort of 179 plasma samples from colorectal cancer (CRC) patients, adenoma patients, and healthy controls, the sensitivity and specificity of the mqMSP assay were 84.9% and 83.3%, respectively. In a head-to-head comparative study, the mqMSP assay also performed better for detecting early-stage (stage I and II) and premalignant polyps than a published SEPT9 assay. In an independent longitudinal cohort of 182 plasma samples (preoperative, postoperative, and follow-up) from 82 CRC patients, the mqMSP assay detected ctDNA in 73 (89.0%) of the preoperative plasma samples. Postoperative detection of ctDNA (within 2 wk of surgery) identified 11 of the 20 recurrence patients and was associated with poorer recurrence-free survival (hazard ratio, 4.20; P = 0.0005). With subsequent longitudinal monitoring, 14 patients (70%) had detectable ctDNA before recurrence, with a median lead time of 8.0 mo earlier than seen with radiologic imaging. The mqMSP assay is cost-effective and easily implementable for routine clinical monitoring of CRC recurrence, which can lead to better patient management after surgery.
23. A systematic evaluation of stool DNA preparation protocols for colorectal cancer screening via analysis of DNA methylation biomarkers. Shengnan Jin, Qian Ye, Yanping Hong, Wenqing Dai, Chengliang Zhang, Weihao Liu, Ying Guo, Dewen Zhu, Zhengzheng Zhang, Shiliang Chen, Yourong Wang, Dandan Li, Wen Ma, Zhengquan Yang, Jinlei Li, Zhihai Zheng, Ju Luan, Xiaoli Wu, Feizhao Jiang, Chang Xu and Chunming Ding. Clinical Chemistry and Laboratory Medicine (CCLM). 2021; 59(1): 91–99
 Objectives
Objectives
Colorectal cancer (CRC) screening using stool samples is now in routine use where tumor DNA methylation analysis for leading markers such as NDRG4 and SDC2 is an integral part of the test. However, processing stool samples for reproducible and efficient extraction of human genomic DNA remains a bottleneck for further research into better biomarkers and assays.
Methods
We systematically evaluated several factors involved in the processing of stool samples and extraction of DNA. These factors include: stool processing (solid and homogenized samples), preparation of DNA from supernatant and pellets, and DNA extraction with column and magnetic beads-based methods. Furthermore, SDC2 and NDRG4 methylation levels were used to evaluate the clinical performance of the optimal protocol.
Results
The yield of total and human genomic DNA (hgDNA) was not reproducible when solid stool scraping is used, possibly due to sampling variations. More reproducible results were obtained from homogenized stool samples. Magnetic beads-based DNA extraction using the supernatant from the homogenized stool was chosen for further analysis due to better reproducibility, higher hgDNA yield, lower non-hgDNA background, and the potential for automation. With this protocol, a combination of SDC2 and NDRG4 methylation signals with a linear regression model achieved a sensitivity and specificity of 81.82 and 93.75%, respectively.
Conclusions
Through the systematic evaluation of different stool processing and DNA extraction methods, we established a reproducible protocol for analyzing tumor DNA methylation markers in stool samples for colorectal cancer screening.
(Methods section) For magnetic beads-based method, Apostle Stool gDNA Isolation Kit (APOSTLE) was used according to the manufacturer’s instructions. Either 0.2 g pellets or 0.2 mL supernatant from homogenized stool was mixed with 1 mL lysis buffer (APOSTLE) for DNA extractions.
22. Electronic Cigarettes Induce Mitochondrial DNA Damage and Trigger TLR9 (Toll-Like Receptor 9)-Mediated Atherosclerosis. Jieliang Li , Luong Huynh , William D. Cornwell , Moon-Shong Tang , Hannah Simborio , Jing Huang , Beata Kosmider , Thomas J. Rogers , Huaqing Zhao , Michael B. Steinberg , Le Thu Thi Le , Lanjing Zhang , Kien Pham , Chen Liu , He Wang. Arteriosclerosis, Thrombosis, and Vascular Biology. 2021;41:839–853
 Objective:
Objective:
Electronic cigarette (e-cig) use has recently been implicated in promoting atherosclerosis. In this study, we aimed to investigate the mechanism of e-cig exposure accelerated atherosclerotic lesion development.
Approach and Results:
Eight-week-old ApoE−/− mice fed normal laboratory diet were exposed to e-cig vapor (ECV) for 2 hours/day, 5 days/week for 16 weeks. We found that ECV exposure significantly induced atherosclerotic lesions as examined by Oil Red O staining and greatly upregulated TLR9 (toll-like receptor 9) expression in classical monocytes and in the atherosclerotic plaques, which the latter was corroborated by enhanced TLR9 expression in human femoral artery atherosclerotic plaques from e-cig smokers. Intriguingly, we found a significant increase of oxidative mitochondria DNA lesion in the plasma of ECV-exposed mice. Administration of TLR9 antagonist before ECV exposure not only alleviated atherosclerosis and the upregulation of TLR9 in plaques but also attenuated the increase of plasma levels of inflammatory cytokines, reduced the plaque accumulation of lipid and macrophages, and decreased the frequency of blood CCR2+ (C-C chemokine receptor type 2) classical monocytes. Surprisingly, we found that cytoplasmic mitochondrial DNA isolated from ECV extract-treated macrophages can enhance TLR9 activation in reporter cells and the induction of inflammatory cytokine could be suppressed by TLR9 inhibitor in macrophages.
Conclusions:
E-cig increases level of damaged mitochondrial DNA in circulating blood and induces the expression of TLR9, which elevate the expression of proinflammatory cytokines in monocyte/macrophage and consequently lead to atherosclerosis. Our results raise the possibility that intervention of TLR9 activation is a potential pharmacological target of ECV-related inflammation and cardiovascular diseases.
21. Liquid Biopsy: Current Status and Future Directions. Front Line Genomics. Jan 2021.
 Case study: a new scalable and automatable method for the extraction of cfDNA.
Case study: a new scalable and automatable method for the extraction of cfDNA.
Liquid biopsies represent a promising area of cancer testing as taking blood is less invasive than tumor biopsies. The cell free DNA (cfDNA) present in the blood includes DNA derived from cancer cells and cancer biomarkers can be detected in the extracted cfDNA. Whole blood also contains genomic DNA, and can be removed by centrifugation, resulting in plasma. cfDNA is present in very small amounts in blood or plasma, and thus larger amounts of plasma are required for many applications. Larger extractions are more challenging to automate, as they require additional pipetting steps. Here we present a novel cfDNA extraction kit and show its compatibility with extractions from 200 μl – 5 mL. We discuss the optimisation of the method and demonstrate automation on a KingFisher Duo. This workflow can also be automated on a Biomek i7 Automated Workstation. We demonstrate that this kit can be used for NGS and produce results comparable to other commercial kits.
Publications (2020)
20. Apostle COVID-19 RNA Extraction System Applied in the Effective Detection of SARS-CoV-2. Horner S. Apostle Inc. Application Note. Oct 2020.
The current coronavirus disease 2019 (COVID-19) pandemic started in late 2019. COVID-19 is the result of severe acute respiratory syndrome 2 (SARS-CoV-2) virus contraction. COVID-19 is often accompanied by a wide range of symptoms including fever, cough, and shortness of breath. The SARS-CoV-2 virus consists of a ~30 kb RNA genome encoding for 15 proteins, including the spike protein that enables the virus to enter host cells. The current gold standard qualitative detection method, qRT-PCR, reverse transcribes the viral RNA into cDNA, which is subsequently amplified and quantitated. This application note illustrates the effective detection of SARS-CoV-2 using the Apostle COVID-19 Viral RNA Isolation Automation System and qRT-PCR in clinical lab settings. This system uses efficient magnetic nanoparticle technology for fast extraction and purification of viral nucleic acids from various types of biological samples collected in transport media. The proficient and consistent systems provide reliable test results to individuals that contribute to COVID-19 pandemic relief.
The use of Apostle COVID-19 Viral RNA Isolation Automation System has proven to be an effective process as a part in detecting SARS-CoV-2 from more than two million samples carried out by our clients in clinical lab settings.
19. Liquid Biopsies Generate Ample Insights from Sparse Clues. Jon Kelvey Genetic Engineering & Biotechnology News. Vol. 40, No. 8, August 1, 2020; pp. 44–46, 48, 49
 (Note: Apostle technologies are discussed in this news article.)
(Note: Apostle technologies are discussed in this news article.)
Duplex sequencing, target enrichment, and other technologies reveal incipient and residual disease as well early signs of drug resistance. [pdf]
18. High-resolution DNA size enrichment using a magnetic nano-platform and application in non-invasive prenatal testing. Bo Zhang, Shuting Zhao, Hao Wan, Ying Liu, Fei Zhang, Xin Guo, Wenqi Zeng, Haiyan Zhang, Linghua Zeng, Jiale Qu, Ben-Qing Wu, Xinhong Wan, Charles R. Cantor and Dongliang Ge. Analyst. July 2020, 145, 5733-5739
 Precise DNA sizing can boost sequencing efficiency, reduce cost, improve data quality, and even allow sequencing of low-input samples, while current pervasive DNA sizing approaches are incapable of differentiating DNA fragments under 200 bp with high resolution (<20 bp). In non-invasive prenatal testing (NIPT), the size distribution of cell-free fetal DNA in maternal plasma (main peak at 143 bp) is significantly different from that of maternal cell-free DNA (main peak at 166 bp). The current pervasive workflow of NIPT and DNA sizing is unable to take advantage of this 20 bp difference, resulting in sample rejection, test inaccuracy, and restricted clinical utility. Here we report a simple, automatable, high-resolution DNA size enrichment workflow, named MiniEnrich, on a magnetic nano-platform to exploit this 20 bp size difference and to enrich fetal DNA fragments from maternal blood. Two types of magnetic nanoparticles were developed, with one able to filter high-molecular-weight DNA with high resolution and the other able to recover the remaining DNA fragments under the size threshold of interest with >95% yield. Using this method, the average fetal fraction was increased from 13% to 20% after the enrichment, as measured by plasma DNA sequencing. This approach provides a new tool for high-resolution DNA size enrichment under 200 bp, which may improve NIPT accuracy by rescuing rejected non-reportable clinical samples, and enable NIPT earlier in pregnancy. It also has the potential to improve non-invasive screening for fetal monogenic disorders, differentiate tumor-related DNA in liquid biopsy and find more applications in autoimmune disease diagnosis.Two types of magnetic nanoparticles were developed, with one able to filter high-molecular-weight DNA with high resolution and the other able to recover the remaining DNA fragments under the size threshold of interest with >95% yield. Using this method, the average fetal fraction was increased from 13% to 20% after the enrichment, as measured by plasma DNA sequencing. This approach provides a new tool for high-resolution DNA size enrichment under 200 bp, which may improve NIPT accuracy by rescuing rejected non-reportable clinical samples, and enable NIPT earlier in pregnancy. It also has the potential to improve non-invasive screening for fetal monogenic disorders, differentiate tumor-related DNA in liquid biopsy and find more applications in autoimmune disease diagnosis.
Precise DNA sizing can boost sequencing efficiency, reduce cost, improve data quality, and even allow sequencing of low-input samples, while current pervasive DNA sizing approaches are incapable of differentiating DNA fragments under 200 bp with high resolution (<20 bp). In non-invasive prenatal testing (NIPT), the size distribution of cell-free fetal DNA in maternal plasma (main peak at 143 bp) is significantly different from that of maternal cell-free DNA (main peak at 166 bp). The current pervasive workflow of NIPT and DNA sizing is unable to take advantage of this 20 bp difference, resulting in sample rejection, test inaccuracy, and restricted clinical utility. Here we report a simple, automatable, high-resolution DNA size enrichment workflow, named MiniEnrich, on a magnetic nano-platform to exploit this 20 bp size difference and to enrich fetal DNA fragments from maternal blood. Two types of magnetic nanoparticles were developed, with one able to filter high-molecular-weight DNA with high resolution and the other able to recover the remaining DNA fragments under the size threshold of interest with >95% yield. Using this method, the average fetal fraction was increased from 13% to 20% after the enrichment, as measured by plasma DNA sequencing. This approach provides a new tool for high-resolution DNA size enrichment under 200 bp, which may improve NIPT accuracy by rescuing rejected non-reportable clinical samples, and enable NIPT earlier in pregnancy. It also has the potential to improve non-invasive screening for fetal monogenic disorders, differentiate tumor-related DNA in liquid biopsy and find more applications in autoimmune disease diagnosis.Two types of magnetic nanoparticles were developed, with one able to filter high-molecular-weight DNA with high resolution and the other able to recover the remaining DNA fragments under the size threshold of interest with >95% yield. Using this method, the average fetal fraction was increased from 13% to 20% after the enrichment, as measured by plasma DNA sequencing. This approach provides a new tool for high-resolution DNA size enrichment under 200 bp, which may improve NIPT accuracy by rescuing rejected non-reportable clinical samples, and enable NIPT earlier in pregnancy. It also has the potential to improve non-invasive screening for fetal monogenic disorders, differentiate tumor-related DNA in liquid biopsy and find more applications in autoimmune disease diagnosis.
17. Improved conversion in extraction, library construction, and capture improve sensitivity for variants in liquid biopsy samples. Nicole Roseman, Shilpa Parakh, Hsiao-Yun Huang, Kevin Lai, Timothy Barnes, Lyn Lewis, Ushati Das Chakravarty, Anastasia Potts, Alisa Jackson, Amy Yoder, Jessica Sheu, Tzu-Chun Chen. In: Proceedings of the Annual Meeting of the American Association for Cancer Research 2020; 2020 Apr 27-28 and Jun 22-24. Philadelphia (PA): AACR; Cancer Res 2020;80(16 Suppl):Abstract nr 5863.
Demonstrate performance of a complete automation and reagent workflow for analysis of cfDNA from bodily fluids. The efficient extraction of cfDNA from bodily fluids is a unique challenge due to the very low concentrations of nucleic acid. The extraction process along with library preparation is a laborious workflow, where human variability can lead to increased variability in the downstream analysis. Integrated DNA Technology (IDT) and Beckman Coulter (BC) have teamed up to provide a complete automation and reagent workflow for analysis of low frequency variants in cfDNA. The Apostle MiniMax™ High Efficiency Isolation Kit from BC provides complex, utilized magnetic nanoparticles to effectively capture cfDNA. IDT's library prep kit utilizes novel chemistry to maximize conversion, suppress adapter-dimer formation, reduce chimera rates, and facilitate double strand consensus analysis to call ultra-low frequency variants. Finally, IDT's xGen™ hybrid capture products maintain high library diversity and on-target rates to enable low frequency variant calling regardless of panel size. The Biomek i5 and i7 Hybrid workstations bring out the best performance from these reagents. The Biomek NGS workstations protocol is written with a modular design with safe stop points, making it customizable for each lab. The automated protocol uses Beckman's Demonstrated Method Interface tools which include: Biomek Method Launcher to run the method without going into Biomek software, Method Options Selector to choose the run parameters with a user friendly interface, Guided labware Setup to set the deck with labware based on the run parameters, DeckOptix Final Check software to help reduce deck setup errors. We demonstrate the performance of this complete workflow with a range of plasma inputs (4-8 mL). Using control samples with known variant frequencies, the workflow yields high library complexity, 100% positive predictive value, and reliable detection of <0.5% mutant allele frequency variants. With real cfDNA, the workflow demonstrates both high cfDNA and sequencing library yields along with high library complexity. The combination of these reagents on the Biomek workstations provides a robust and reproducible solution for the analysis of cfDNA.
16. G3viz: an R package to interactively visualize genetic mutation data using a lollipop-diagram. Xin G, Bo Z, Wenqi Z, et al. Bioinformatics. 2020; 36(3):928–929
 The lollipop-diagram is one of the widely used graphical representations to visualize and explore translational effects of genetic mutations in cancer genomics. However, an easy-to-use lollipop-diagram tool with full functionality is still lacking. Here, we introduce g3viz, an R package that enables researchers to explore genetic mutation data using a lollipop-diagram in a web browser. With a few lines of R code, users can interactively visualize data details, annotate findings and export resultant diagrams in high-quality figures. Because of usefulness and usability, g3viz can be generally exploited by researchers with different levels of bioinformatics skills and programming experience.
The lollipop-diagram is one of the widely used graphical representations to visualize and explore translational effects of genetic mutations in cancer genomics. However, an easy-to-use lollipop-diagram tool with full functionality is still lacking. Here, we introduce g3viz, an R package that enables researchers to explore genetic mutation data using a lollipop-diagram in a web browser. With a few lines of R code, users can interactively visualize data details, annotate findings and export resultant diagrams in high-quality figures. Because of usefulness and usability, g3viz can be generally exploited by researchers with different levels of bioinformatics skills and programming experience.
15. An end-to-end automated workflow for low-frequency variant detection in cfDNA. Tzu-Chun Chen, Nicole Roseman, Han Wei, et al. ASHG. Oct 27-30, 2020
Introduction
Liquid biopsy, as a tool to detect tumor-associated variants in cell-free DNA (cfDNA) from plasma, is a powerful non-invasive approach for biomarker discovery in oncology research. Integrated DNA Technologies (IDT) and Beckman Coulter Life Sciences (BECLS) have teamed up to provide a comprehensive end-to-end automated solution on the Biomek workstations for low frequency variant detection in cfDNA using next generation sequencing. The combined workflow including reagents for cfDNA extraction from plasma, NGS library preparation and hybrid capture, provides a robust and reproducible solution analyzing cfDNA samples.
Conclusions
• We provide an end-to-end automation and reagent workflow from plasma to cfDNA analysis that includes Apostle Minimax, xGenPrism DNA Library Prep Kit, and xGen hybridization capture.
• Apostle MiniMax High Efficiency Isolation Kit provides high yield extraction of cfDNA. • The xGen Prism DNA library preparation kit, optimized for low input and degraded
samples such as cfDNA, provides sensitive and accurate detection of ultra-low frequency variants.
14. A complete automation and reagent workflow for analysis of cfDNA: from plasma to variants. Roseman N, Parakh S, Lai K, Sheu J, Wei H, Niccum B, Chen T, Huang H, Barnes T, Lewis L, Chakravarty UD, and Potts A. Advances in Genome Biology and Technology (AGBT). (abstract #511). Marco Island, FL. Feb 23-26, 2020
 As cost of sequencing continues to drop, the throughput and complexity of NGS assays have risen precipitously. At the same time, the types of samples being used for these assays have expanded as researchers find value in studying low input, degraded biological samples, such as cell free DNA (cfDNA). However, efficient extraction of cfDNA from bodily fluids is challenging due to the very low concentrations of nucleic acid. Manual protocols involved in the NGS workflow – DNA extraction, library preparation, and target enrichment – are often laborious and require significant hands on time, which can increase variability in the downstream analysis. Thus, complete automation workflows can increase throughput, reduce hands-on time, and minimize error. Integrated DNA Technologies (IDT) and Beckman Coulter (BC) have teamed up to provide a complete automation and reagent workflow for analysis of low frequency variants in cfDNA. The Apostle MiniMaxTM High Efficiency Isolation Kit from BC is a magnetic nanoparticle based kit that extracts cfDNA from all bodily fluids. IDT’s xGenTM Prism DNA library prep kit provides novel chemistry to maximize conversion, suppress adapter-dimer formation, reduce chimera rates, and facilitate UMI-based error correction to call ultra-low frequency variants. IDT’s xGenTM target enrichment products maintain high library diversity and on-target rates to enable low frequency variant calling regardless of panel size. The combination of these reagents on the Biomek workstations provides a robust and reproducible solution for the analysis of cfDNA.
As cost of sequencing continues to drop, the throughput and complexity of NGS assays have risen precipitously. At the same time, the types of samples being used for these assays have expanded as researchers find value in studying low input, degraded biological samples, such as cell free DNA (cfDNA). However, efficient extraction of cfDNA from bodily fluids is challenging due to the very low concentrations of nucleic acid. Manual protocols involved in the NGS workflow – DNA extraction, library preparation, and target enrichment – are often laborious and require significant hands on time, which can increase variability in the downstream analysis. Thus, complete automation workflows can increase throughput, reduce hands-on time, and minimize error. Integrated DNA Technologies (IDT) and Beckman Coulter (BC) have teamed up to provide a complete automation and reagent workflow for analysis of low frequency variants in cfDNA. The Apostle MiniMaxTM High Efficiency Isolation Kit from BC is a magnetic nanoparticle based kit that extracts cfDNA from all bodily fluids. IDT’s xGenTM Prism DNA library prep kit provides novel chemistry to maximize conversion, suppress adapter-dimer formation, reduce chimera rates, and facilitate UMI-based error correction to call ultra-low frequency variants. IDT’s xGenTM target enrichment products maintain high library diversity and on-target rates to enable low frequency variant calling regardless of panel size. The combination of these reagents on the Biomek workstations provides a robust and reproducible solution for the analysis of cfDNA.
Conclusion
• Apostle Minimax, xGen Prism, and xGen hybridization capture provide a complete automated solution from plasma to sequencing on the Beckman Coulter Biomek platforms
• Apostle Minimax provides higher quantity and quality cfDNA extraction compared to a competitor kit
• xGen Prism is specifically designed for low input, degraded samples, such as cfDNA and generates higher coverage and complexity compared to competitor kits
• UMI-based error correction can be used to eliminate nearly all false positive calls and enables accurate low frequency variant calling
13. cfDNA Extraction Efficiency Affects NGS Data. Han Wei, Ph.D., Beckman Coulter Life Sciences. Indianapolis, IN. Technical Note 2020.
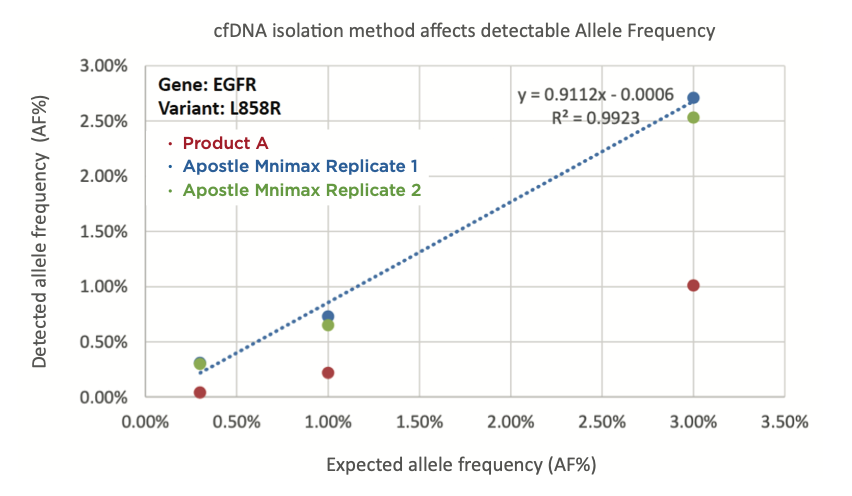
Figure 1. cfDNA extraction method affects allele frequency (AF%). EGFR L858R standards with AF% of 0.3%, 1.0%, and 3.0% were spiked into plasma and isolated using Product A (red) or Apostle MiniMax cfDNA isolation kit (blue and green). The eluates were analyzed by NGS (performed by the Institute B) following standard protocol and AF% calculated using their established workflow. cfDNA isolated by Apostle MiniMax cfDNA isolation kit shows higher concordance between detected AF% and expected AF%.
In order to understand how cfDNA extraction efficiency affects NGS data, we present Institute B titration data. EGFR L858R standards with an allele frequency (AF%) of 0.3%, 1.0%, and 3.0% were spiked into plasma and isolated using either Apostle MiniMaxTM High Efficiency cfDNA Isolation or Product A. The cfDNA was analyzed via NGS and AF% was calculated by Institute B. cfDNA isolated by Apostle MiniMax cfDNA isolation kit shows concordance between detected AF% and expected AF%.
For cancer samples, the allele frequency represents the percentage of sequence reads carrying a mutant allele of an individual patient’s cancer. Researchers often set thresholds for defining the variant allele frequency as mutation; therefore, low cfDNA recovery efficiency can decrease sensitivity and lead to an increase in false negatives for mutation calling.
Publications (2019)
12. Correlation between mutations found in FFPE tumor tissue and paired cfDNA samples. (n=8) Niccum B., Saunders L., Hur A.,Patel A. The American Society of Human Genetics (ASHG). (abstract #1766). Houston, TX. Oct 15, 2019
Liquid biopsies represent a promising area of facilitating cancer research as taking blood is less invasive than tumor biopsies. Cell-free DNA (cfDNA) consists of small (150 – 500 bp) DNA fragments that circulate in the blood. Levels of cfDNA tend to be low in healthy, non-pregnant patients and increased in patients with cancer, pregnancy, or extensive tissue damage. cfDNA is believed to be derived mostly from apoptotic cells and a source for biomarkers for a variety of diseases.
As a non-invasive way to detect disease cfDNA is extracted from blood; however, there is some concern that cfDNA does not contain the same biomarkers as tumor tissue. Tumor tissue is typically removed and stored as formalin-fixed, paraffin-embedded tissue, a process that preserves the morphological structures well but chemically modifies and degrades the nucleic acids.
Despite the difficulties, FFPE tissue (shown on the left) is often used to look for cancer-associated mutations; however it does not always correlate with the mutations seen in cfDNA. In this poster we present a comparison of matched FFPE and plasma samples to determine how many mutation are seen in both tissues. We also look at where the mutational mismatches appear in the chromosome. We found chromosomal regions have different mismatch rates, and we use this to draw conclusions about the best chromosomal locations for biomarkers. We also look at the different results that can come from using multiple different panels.
Here we show that sequencing of cfDNA captures the majority of variants that are found when sequencing FFPE DNA. One result shown here is that more indels are identified using cfDNA than with FFPE DNA. This is especially shown here with breast tissue, while this is inconclusive with lung tissue. More samples should be tested before any conclusions can be derived.
Another interesting finding is the distribution of variants across the genome. There is some indication that variants sequenced using cfDNA is correlated better to the variants found with either cfDNA or FFPE. This result could be used to better understand if there is bias that occurs when sequencing FFPE DNA and where this bias could be from such as cross-linking could be more apparent in parts of chromosomes.
11. Comparison between mutation profiles of paired whole blood and cfDNA samples. Patel A., Saunders L., Hur A. The American Society of Human Genetics (ASHG). (abstract #1767). Houston, TX. Oct 15, 2019
Liquid biopsies are increasingly becoming a tool of choice for researching cancer detection and monitoring. Cell-free DNA or cfDNA is simply small fragments of DNA circulating in bodily fluids. It is also known as circulating cell-free DNA (ccfDNA), circulating tumor DNA (ctDNA) and cell free-fetal DNA (cffDNA). Next-Gen Sequencing of cfDNA is coming into maturity as a non-invasive method to identify mutational profiles in many cancer types.
A big question is how do you separate a germ line variant from a tumor variant. Understanding the difference in a patient sample can give a more thorough understanding of a variant that can be used to study a cancer type. An easy solution is to compare germ line variants from whole blood genomic DNA (gDNA). This would couple easily with plasma sample collection as plasma can be directly separated from a single sample point.
Here we describe a simple method to isolate both gDNA and cfDNA from a donor blood sample and discuss the automation of both extractions. We show the efficacy of cfDNA as reliable biomarker analysis tool by comparing mutations in cfDNA vs whole blood. The study determines if the difference between tumor and germ line mutations can be established and the limitations.
Due to larger volumes necessary to extract sufficient concentrations of cfDNA, automation can assist in the extraction. The Apostle MiniMax™ High Efficiency Cell-Free DNA (cfDNA) extraction kit was automated on the Biomek i-Series. It provides equal recovery of cfDNA as a manual extraction with much less hands on-time. The kit used in the study to extract whole blood, GenFind V3, has also been automated on the Biomek i-Series; allowing for reduced hands with the same quality results as a manual extraction.
Conclusions
• Variants found only in cfDNA could be used as an initial screen for ctDNA analysis
• Apostle MiniMax™ and GenFind V3 can be used together to get a picture of germ line variants and cfDNA, potential ctDNA, variants
• These results show that a holistic view of a cancer subject can be gained by using one sample source, whole blood
10. Correlation between mutations found in FFPE tumor tissue and paired cfDNA samples. Niccum B., Heath C., Saunders L., Hur A.,Patel A. Association for Molecular Pathology (AMP). (abstract #ST103). Baltimore, MD. November 7-9, 2019
Liquid biopsies represent a promising area of facilitating cancer research as blood collection is less invasive than tumor biopsies. Cell free DNA (cfDNA) consists of small (150 – 500 bp) DNA fragments that circulate in the blood. cfDNA levels tend to be low in healthy, non-pregnant patients, and increase in patients with cancer, pregnancy, or extensive damage to tissue. cfDNA is believed to be derive mostly from apoptotic cells for which biomarkers for a variety of diseases have been found in cfDNA.
FFPE tissue is often used to look for cancer-associated mutations despite invasiveness; however it does not always correlate with the mutations seen in cfDNA. In this poster we present a comparison of matched FFPE and plasma samples to determine how many mutation are seen in both tissues. We also look at where the mutational mismatches appear in the chromosome. Different chromosomal regions can have different mismatch rates, and we use this to draw conclusions about the best chromosomal locations for biomarkers. We automated from extraction through sequencing in collaboration with Swift biosciences.
As cfDNA is extracted from blood, it is a non-invasive way to detect disease; however, there is some concern that cfDNA does not contain the same biomarkers as tumor tissue. Tumor tissue is typically removed and stored as formalin-fixed, paraffinembedded tissue, a process that preserves the morphological structures well but chemically modifies and degrades the nucleic acids.
Here we show:
• Sequencing of cfDNA captures the majority of variants that found in sequenced FFPE DNA
• More indels are identified using cfDNA than with FFPE DNA, especially with breast tissue
• Distribution of variants across the genome differs when sequencing FFPE DNA
• More previously identified clinically relevant variants, as identified by the ClinVar database were found when sequencing FFPE DNA
This study is small and further work should be done using larger data sets to gain more conclusive information.
9. Dynamics of Plasma EGFR T790M Mutation in Advanced NSCLC: A Multicenter Study. Yang et al. Targeted Oncology. 2019;14:719-728. Published: 06 November 2019.
(Note: Apostle MiniMax technology is used in this clinical study.)
 Background Droplet digital polymerase chain reaction (ddPCR) is an emerging technology for quantitative cell-free DNA oncology applications. However, a ddPCR assay for the epidermal growth factor receptor (EGFR) p.Thr790Met (T790M) mutation suitable for clinical use remains to be established with analytical and clinical validations. Objective We aimed to develop and validate a new ddPCR assay to quantify the T790M mutation in plasma for monitoring and predicting the progression of advanced non-small-cell lung cancer (NSCLC). Methods Specificity of the ddPCR assay was evaluated with genomic DNA samples from healthy individuals. The inter- and intraday variations of the assay were evaluated using mixtures of plasmid DNA containing wild-type EGFR and T790M mutation sequences. We assessed the clinical utility of the T790M assay in a multicenter prospective study in patients with advanced NSCLC receiving tyrosine kinase inhibitor (TKI) treatment by analyzing longitudinal plasma DNA samples. Results We set the criteria for a positive call when the following conditions were satisfied: (1) T790M mutation frequency > 0.098% (3 standard deviations above the background signal); (2) at least two positive droplets in duplicate ddPCR reactions. Among the 62 patients with advanced NSCLC exhibiting resistance to TKI treatment, 15 had one or more serial plasma samples that tested positive for T790M. T790M mutation was detected in the plasma as early as 205 days (median 95 days) before disease progression, determined by imaging analysis. Plasma T790M concentrations also correlated with intervention after disease progression. Conclusions We developed a ddPCR assay to quantify the T790M mutation in plasma. Quantification of longitudinal plasma T790M mutation may allow noninvasive assessment of drug resistance and guide follow-up treatment in TKI-treated patients with NSCLC. Trial Registration Clinical Trials.gov identifier: NCT02804100.
Background Droplet digital polymerase chain reaction (ddPCR) is an emerging technology for quantitative cell-free DNA oncology applications. However, a ddPCR assay for the epidermal growth factor receptor (EGFR) p.Thr790Met (T790M) mutation suitable for clinical use remains to be established with analytical and clinical validations. Objective We aimed to develop and validate a new ddPCR assay to quantify the T790M mutation in plasma for monitoring and predicting the progression of advanced non-small-cell lung cancer (NSCLC). Methods Specificity of the ddPCR assay was evaluated with genomic DNA samples from healthy individuals. The inter- and intraday variations of the assay were evaluated using mixtures of plasmid DNA containing wild-type EGFR and T790M mutation sequences. We assessed the clinical utility of the T790M assay in a multicenter prospective study in patients with advanced NSCLC receiving tyrosine kinase inhibitor (TKI) treatment by analyzing longitudinal plasma DNA samples. Results We set the criteria for a positive call when the following conditions were satisfied: (1) T790M mutation frequency > 0.098% (3 standard deviations above the background signal); (2) at least two positive droplets in duplicate ddPCR reactions. Among the 62 patients with advanced NSCLC exhibiting resistance to TKI treatment, 15 had one or more serial plasma samples that tested positive for T790M. T790M mutation was detected in the plasma as early as 205 days (median 95 days) before disease progression, determined by imaging analysis. Plasma T790M concentrations also correlated with intervention after disease progression. Conclusions We developed a ddPCR assay to quantify the T790M mutation in plasma. Quantification of longitudinal plasma T790M mutation may allow noninvasive assessment of drug resistance and guide follow-up treatment in TKI-treated patients with NSCLC. Trial Registration Clinical Trials.gov identifier: NCT02804100.
8. Isolation of cell-free DNA (cfDNA) from plasma using Apostle MiniMaxTM High Efficiency cfDNA Isolation kit—comparison of fully automated, semi-automated and manual workflow processing. Brittany Niccum, PhD., Randy Pares and Antonia Hur. Beckman Coulter Life Sciences. Application Note. Sept 2019.
Cell-free DNA is present in plasma, urine, and other bodily fluids. Typically cfDNA is at low concentration and comprises double-stranded DNA fragments that are overwhelmingly short (140 to 180 base pairs). Its small size and low quantities present a challenge for sequencing and other downstream applications due to insufficient cfDNA yield. Subsequently, resulting in a need to extract from more substantial volumes of bodily fluid; yet higher input volumes can be more challenging to manage for high-throughput sample processing.
This application note compares workflows and yield for the extraction of cfDNA using Apostle MiniMax High Efficiency cfDNA Isolation Kit, following the manual protocol, automating the extraction using the Biomek i7 Hybrid Workstation, and semi-automating the extraction using the KingFisher Duo Prime Sample Purification System. These potential solutions help mitigate some of the challenges of processing large volume samples. Automating the chemistry can also reduce the risk of human error and reduce hands-on time, therefore giving the user the ability to run more samples in a day.
7. Cell-Free DNA Isolation Kit. Science. 17 May 2019:Vol. 364, Issue 6441, pp. 696. DOI: 10.1126/science.364.6441.696-a. (Featured in New Products section)
Surpassing industry standards for yield and purity of cell-free DNA (cfDNA), Apostle MiniMax from Beckman Coulter is a magnetic nanoparticle–based kit that extracts cfDNA from plasma using manual or automated workflows. Due to limited cfDNA concentration, sufficient yield for NGS or other downstream applications can require higher volumes of plasma. As the volume increases, issues such as recovery efficiency and workflow complexity often arise. Plasma contains a host of contaminants that are problematic at this scale and can reduce assay sensitivity. Apostle MiniMax technology performs reliably across a range of volume inputs, consistently recovering high quantities of cfDNA while effectively removing contaminants.
6. A workflow for medium-throughput isolation of cfDNA from plasma samples using Apostle MiniMaxTM on the KingFisherTM Technology. Brittany Niccum, PhD. Beckman Coulter Life Sciences. Application Note. 2019.
Cell-free DNA (cfDNA) is found in various bodily fluids. cfDNA has a characteristic size of approximately 175 bp. Due to its small size and low quantity the main challenge is to get enough cfDNA for sequencing. Subsequently, this results in a need to extract from larger volumes of bodily fluid, yet higher input volumes can be more difficult to manage for high-throughput sample processing.
This application note demonstrates the use of Apostle MiniMax High Efficiency cfDNA Isolation Kit, in conjunction with the KingFisher Duo Prime automated protein purification system. This potential solution that mitigates some of the challenges of processing large volume samples. Automating the chemistry can also reduce the risk of human error, reduce hands-on time and total time; therefore giving the user the ability to run more samples in a day.
5. Biomek Automated Genomic Sample Prep Accelerates Research: Biomek i-Series Automation of the Apostle MiniMaxTM High Efficiency cfDNA Isolation Kit. Application Overview. Beckman Coulter Life Sciences. 2019
The Apostle MiniMax High Efficiency cfDNA Isolation Kit isolates cell free DNA (cfDNA) and circulating tumor DNA (ctDNA) from plasma collected from blood collection tubes containing EDTA and other blood collection tube types as well as from serum and urine. The kit produces high quality extracted cfDNA that can be used in downstream genomic assays, such as PCR amplification and next-generation sequencing. Proteins in cell free plasma are digested and cfDNA is captured using Apostle’s proprietary technology. Contaminants are removed from the samples through several simple washes, leaving high quality cfDNA samples that are ready for elution. In this technical note, we demonstrate the automated performance of the Apostle MiniMax High Efficiency cfDNA Isolation Kit on the Biomek i7 Hybrid Genomics Workstation.
When compared to manual operations, the Apostle MiniMax High Efficiency cfDNA Isolation Kit automated on Biomek platforms provide:
• Reduced hands-on-time and increased throughput
• Option to run the method end-to-end with only setup and tear-down touch points
• Reduction in pipetting errors
• Standardized workflow for improved results
• Quick implementation with demonstrated methods
• Knowledgeable support for reagents, automation and methods all from a single vendor
4. Correlation between mutations found in FFPE tumor tissue and paired cfDNA samples. (n=3) Saunders L and Patel A. American Association for Cancer Research (AACR). (abstract #2233). Atlanta, GA. April 2, 2019
Liquid biopsies represent a promising area of facilitating cancer research as taking blood is less invasive than tumor biopsies. The cell free DNA (cfDNA) present in the blood includes DNA derived from cancer cells and cancer biomarkers can be detected in the extracted cfDNA. However, cfDNA is a less direct view of what is happening in the tumor, and can have a different genetic profile than the tumor tissue itself.
Tumor tissue is typically removed and stored as formalin-fixed, paraffin-embedded tissue, a process that preserves the morphological structures well but chemically modifies and degrades the nucleic acids. This tissue is often used to look for cancer- associated mutations despite these difficulties; however, it does not always correlate with the mutations seen in cfDNA.
In this poster we present a comparison of matched FFPE and plasma samples to determine how many mutations are seen in both tissues. We also look at where the mutational mismatches appear in the chromosome. Different chromosomal regions can have different mismatch rates, and we use this to draw conclusions about the best chromosomal locations for biomarkers.
Conclusions:
Both FFPE and cfDNA detect at least two thirds of the observed indels and at least 90% of the observed SNVs. As SNVs are more likely to be found in both tissue types, they are more suitable for biomarker use if looking across different tissues. This was true for all three cancers tested.
Different regions of the chromosome have different rates of mismatches in mutation detection between plasma and FFPE tissue. The first 10-30%, 40-50%, 60-80%, and 90-100% of the chromosome have the lowest rates of mismatch and provide the best locations for biomarkers detectible in both tissues. Future work will focus in increasing the sample size to further narrow down the areas of the chromosome with the highest likelihood of good mutation detection in both plasma and FFPE.
3. A new Scalable and automatable method for the extration of cfDNA. Saunders LP, Hur A, Niccum B, and Patel A. Advances in Genome Biology and Technology (AGBT). (abstract #419). Marco Island, FL. Feb 28, 2019
Liquid biopsies represent a promising area of cancer testing as taking blood is less invasive than tumor biopsies. The cell free DNA (cfDNA) present in the blood includes DNA derived from cancer cells and cancer biomarkers can be detected in the extracted cfDNA.
Whole blood also contains genomic DNA, and can be removed by centrifugation, resulting in plasma. cfDNA is present in very small amounts in blood or plasma, and thus larger amounts of plasma are required for many applications. Larger extractions are more challenging to automate, as they require additional pipetting steps.
Here we present a novel cfDNA extraction kit and show its compatibility with extractions from 200 μl – 5 mL. We discuss the optimization of the method and demonstrate automation on a KingFisher Duo. This workflow can also be automated on a Biomek i7 Automated Workstation. We demonstrate that this kit can be used for NGS and produce results comparable to other commercial kits.
Conclusions:
• DNA can be extracted from 200 μL to 5 mL of plasma
• The Apostle MiniMax kit removes the PCR inhibitors present in plasma
• Genomic contamination is not present in the extracted cfDNA
• Extraction of 1 mL plasma can be automated on a KingFisher instrument with yields similar to manual extraction
• Similar numbers of mutations were found in cancer plasma with the Apostle MiniMax kit and another commercial kit.
The Apostle MiniMax kit is a versatile new cfDNA kit that can extract from a wide range of sample amounts and be run either manually or on a variety of automation systems.
2. cfDNA Extraction from Plasma for Liquid Biopsy: Apostle MiniMaxTM High Efficiency cfDNA Isolation Kit. Beckman Coulter Life Sciences, Data Sheet. 2019.
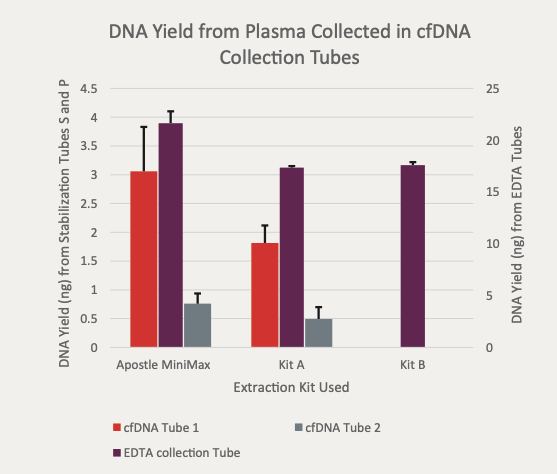
(Figure 1) "For each tube the Apostle MiniMax extracted higher total yield of DNA. "
Apostle MiniMax High Efficiency cfDNA Isolation Kit, Apostle MiniMax is a cell–free DNA (cfDNA) isolation reagent kit, built on magnetic bead–based technology. Apostle MiniMax has been demonstrated to purify cfDNA from human plasma in both manual and automated workflows.
• Data representative of results of cfDNA extracted from 1–5mL of plasma
• Demonstrated compatibility with a variety of collection tubes
• cfDNA purity shown to be suitable for downstream PCR based assays
Publications (2018)
1. Competitive evolution of NSCLC tumor clones and the drug resistance mechanism of first-generation EGFR-TKIs in Chinese NSCLC patients. Deng et al. Heliyon. VOLUME 4, ISSUE 12, E01031, DECEMBER 01, 2018
Purpose - Although many studies have reported on the resistance mechanism of first-generation EGFR TKIs (1st EGFR TKIs) treatment, large-scale dynamic ctDNA mutation analysis based on liquid biopsy for non-small cell lung cancer (NSCLC) in the Chinese population is rare. Using in-depth integration and analysis of ctDNA genomic mutation data and clinical data at multiple time points during the treatment of 53 NSCLC patients, we described the resistance mechanisms of 1st EGFR TKIs treatment more comprehensively and dynamically. The resulting profile of the polyclonal competitive evolution of the tumor provides some new insights into the precise treatment of NSCLC.
Experimental design - A prospective study was conducted in patients with advanced NSCLC with acquired resistance to erlotinib, gefitinib or icotinib. By liquid biopsy, we detected mutations in 124 tumor-associated genes in the context of drug resistance. These 124 genes covered all tumor therapeutic targets and related biological pathways. During the entire course of treatment, the interval between two liquid biopsies was two months.
Results - Unlike the common mutations tested in tissue samples, our data showed a higher coverage of tumor heterogeneity (32.65%), more complex patterns of resistance and some new resistance mutation sites, such as EGFR p.V769M and KRAS p.A11V. The major resistance-associated mutations detected were still EGFR p.T790M (45.28%), other point mutations in EGFR (33.9%), and KRAS and NRAS mutations (15.09%). These mutation ratios might be considered as a preliminary summary of the characteristics of Chinese patients. In addition, starting from the two baseline mutations of the EGFR gene (19del vs. L858R), we first described the detailed mutation profile of the EGFR gene. Although there was no significant difference in the number of patients with EGFR p.19del and EGFR p.L858R baseline mutations (24% vs. 16%, P = 0.15), patients from the EGFR p.19del baseline group were much more likely to develop EGFR p.T790M resistance mutations (62.1% vs. 19.3%, P = 0.007). Through careful integration of gene mutation information and clinical phenotype information, an interesting phenomenon was found. Although the variant allele fraction (VAF) of the EGFR p.T790M mutation was significantly linearly correlated with that of the EGFR drug-sensitive mutation (r = 0.68, P = 0.00025), neither VAF was associated with the tumor volume at the advanced stage. It was shown that other tumor clones might contribute more to the resistance to 1st EGFR TKIs treatment than tumor clones carrying the EGFR p.T790M mutation when resistance developed. By further analysis, we found that, in some patients, when the primary tumor clones detected were those carrying EGFR−/− mutations (both types the EGFR p.19del/p.L858R and EGFR p.T790M mutation types were missing), most of them showed a poor prognosis and ineffective late treatment, indicating that EGFR−/− played a more important role than EGFR p.T790M in the process of NSCLC drug resistance in these patients. From the perspective of the clonal evolution of NSCLC tumor cells, these phenomena could be explained by the competitive evolution between different tumor clones. In addition, two new mutations, KRAS p.A11V and EGFR p.V769M, emerged significantly during drug resistance in NSCLC patients and had shown obvious competitive clonal evolution characteristics. Combined with clear clinical drug resistance phenotypic information, we believed that these two new mutations might be related to new drug resistance mechanisms and deserve further study. We have also seen an interesting phenomenon. In some patients undergoing 1st EGFR TKIs treatment, the EGFR p.T790M mutation appeared, disappeared, and reappeared, and this spatial and temporal diversity of the EGFR p.T790M mutation was regulated by targeted drug and chemotherapy and was correlated with the individual tumor mutation profile.
Conclusions - The constitution and competitive evolution of the tumor clones have a decisive influence on treatment and can be regulated by targeted drugs and chemotherapy. Additionally, EGFR p.T790M spatial and temporal diversity during treatment warrants more attention, and this spatial and temporal diversity may be useful for the choice of treatment strategies for certain NSCLC patients. Through longitudinal cfDNA sample analysis, the resistance mechanism and dynamic clinical features of Chinese NSCLC patients are systematically established as reliable and meaningful to understand acquired resistance and make further personalized treatment decisions dynamically. Two new potential drug resistance-associated mutations in EGFR and KRAS have been found and are worthy of further study. Finally, our research shows that the evolutionary process of tumor cloning can be artificially regulated and intervened, possibly providing a new way to treat tumors.
(Methods section) - Cell free DNA was isolated using Apostle MiniMaxTM High Efficiency cfDNA Isolation Kit (Standard Edition) according to the manufacturer's protocol.
History of Accreditations